J. Jpn. Acad. Midwif., Vol. 25, No. 1, 5-12, 2011
*1 Graduate School of Comprehensive Human Sciences, University of Tsukuba *2 Graduate School of Medicine, Ehime University
Received 18 March 2010; accepted 16 January 2011
Original article
Relationship between sleep-disordered breathing
and perinatal outcome in pregnant women
Sachiyo MIYAGAWA
*1, Yoko EMORI
*1, Atsuko KAWANO
*1,
Susumu SAKURAI
*2, and Takeshi TANIGAWA
*2Abstract Objective
The purpose of this study is to investigate the influence of sleep-disordered breathing (SDB) in pregnant wom-en on birth outcomes.
Methods
The subjects were 179 females after Week 28 of pregnancy who had undergone pregnancy checkups in one obstetric hospital. We calculated the peripheral arterial blood ODI per hour during sleep at night by dividing the frequency at which the oxygen saturation did not reach the reference value by the duration of examination using a pulse oximeter.
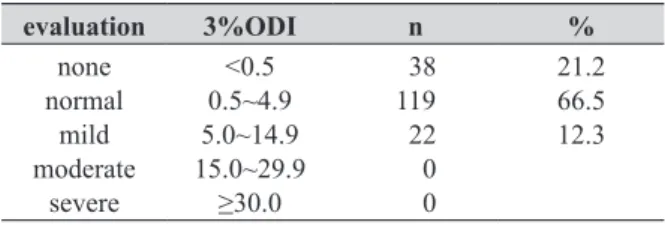
Concerning the criteria, patients with a 3%ODI of less than 0.5 were regarded as showing the absence of SDB, those with values ranging from 0.5 to 4.9 as showing the normal range, those with values ranging from 5 to 14.9 as having mild SDB, those with values ranging from 15 to 29.9 as having moderate SDB, and those with a 3%ODI of 30 or more as having severe SDB.
Multiple logistic regression analysis was performed to assess the birth outcomes associated with SDB. Results
The 3%ODI was less than 0.5 in 38 (21.2%), 0.5 to 4.9 in 119 (66.5%), and 5 to 14.9 in 22 (12.3%). Therefore, we compared a mild SDB group (3%ODI≥5, n=22) with a normal group (3%ODI<5, n=157).
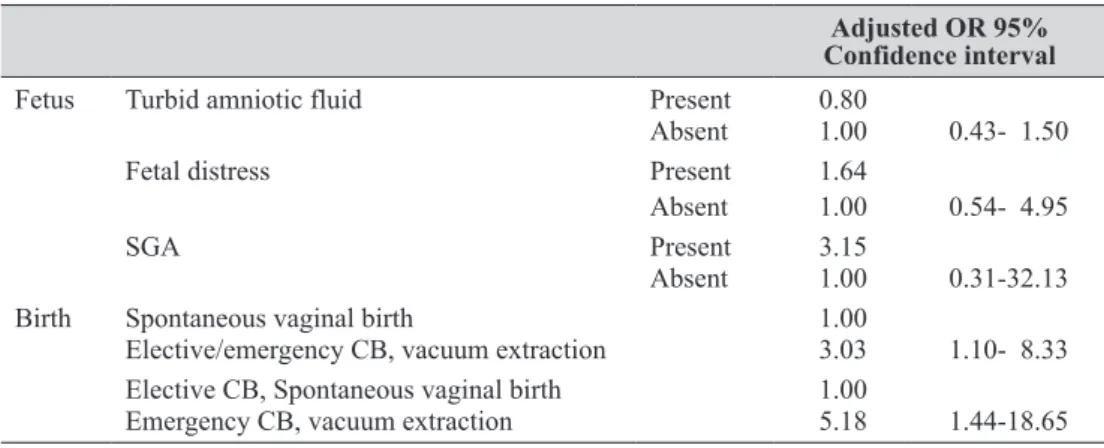
We calculated the adjusted odds ratio (OR), regarding parity and obesity as confounding variables. Regarding spontaneous vaginal birth as the baseline, the adjusted OR of elective or emergency cesarean birth (CB)/vacuum extraction was 3.03 (95% Confidence Interval (CI): 1.10-8.33). Regarding elective CB/spontaneous vaginal birth as the baseline, the adjusted OR of emergency CB/vacuum extraction was 5.18 (95%CI: 1.44-18.65).
Conclusion
Mild SDB in pregnant women suggested that influence to the birth outcomes. Therefore, screening with a pulse-oximeter should be effectively utilized in multiparas with a history of spontaneous abortion and pregnant women complaining of frequent awakening at night or sleep disturbance. In addition, early intervention to improve the state of sleep and treat sleep disturbance may be necessary to promote a safer pregnancy.
Key words: Sleep-disordered breathing (SDB), pregnant women, pulse-oximeter, screening
I. Introduction
In pregnant women, sleep-disordered breathing (SDB), which is characterized by frequent apnea dur-ing sleep and a decrease in the air flow, reduces the fe-tal heart rate, requiring neonafe-tal intervention after birth
(Sleep Disorder Meeting 2005; Inoue & Yamashiro, 2006; Sahin, Koken, Cosar, et al., 2008). However, in Japan, no survey involving pregnant women has inves-tigated SDB, excluding a few case reports on marked SDB. (Shiomi, Yabushita, Wakastuki, 2005). Accord-ing to an international symptom-based survey
regard-9%, respectively (Pien, Fife, Pack, et al., 2005 ; Young, Palta, Dempsey, et al., 1993).
On the other hand, some studies reported that the frequency of sleepiness, a symptom of SDB, increased with the course of pregnancy (Pien, Fife, Pack, et al., 2005 ; Mindell & Jacobson, 2000), whereas others indi-cated that this symptom was frequent in the first trimes-ter (Suzuki, Ohida, Sone, et al., 2003). Pregnant wom-en's sleepiness has not been emphasized as an abnormal sign, assuming that it may be related to physical changes such as the secretion of pregnancy-associated hormones and an increase in the uterine capacity. However, sev-eral studies showed that a sleep time of 6 hours or less for pregnant women prolonged the duration of birth, increasing the rate at which cesarean birth (CB) was se-lected (Suzuki, Ohida, Sone, et al., 2003 ; Shinkawa , Shimada, Fujita, 2008 ; Lee & Gay, 2004), suggesting an association between sleep and the outcome of birth.
In pregnant women with SDB, the body mass index (BMI) is higher than in those without SDB, and the cer-vical circumference is greater (Young, Palta, Dempsey, et al., 1993). In addition, SDB may cause abortion/still-birth (Shiomi, Yabushita, Wakastuki, 2005 ; Shiomi & Yabushita, 2006).
In this study, we investigated the morbidity of SDB in Japanese pregnant women and its influence on birth outcomes.
II. Methods 1. Subjects
The subjects were 184 females after Week 28 of pregnancy who had undergone pregnancy checkups in one obstetric hospital in a prefecture. Of these, 179 were analyzed, excluding 4 in whom this study was discon-tinued and 1 in whom the data were not available.
2. Survey period
From July 2007 until May 2009.
3. Study methods and instruments
For SDB assessment, a pulse-oximeter was
em-1) Pulse-oximeter
Using a PMP-200 pulse-oximeter (Pacific Japan, Inc., Tokyo, Japan), we calculated the peripheral arterial blood oxygen desaturation index (ODI) per hour during sleep at night by dividing the frequency at which the ox-ygen saturation did not reach the reference value by the duration of examination. For example, 3%ODI refers to the frequency at which the oxygen saturation decreases by 3% or more of the baseline per hour. The sensitivity and specificity of assessment with a pulse-oximeter are 80 and 95% of those of polygraphy, respectively (Naka-mata, Kubota, Sakaki, 2003). Concerning the criteria, patients with a 3%ODI of less than 0.5 were regarded as showing the absence of SDB, those with values ranging from 0.5 to 4.9 as showing the normal range, those with values ranging from 5 to 14.9 as having mild SDB, those with values ranging from 15 to 29.9 as having moderate SDB, and those with a 3%ODI of 30 or more as having severe SDB. In this survey, patients with mild to severe SDB (3%ODI≥5) were assigned to a group with SDB, and those with values within the normal range or with-out SDB (3%ODI<5) to a group withwith-out SDB.
2) Questionnaire
We investigated the interval until sleep induction (min), total sleep time (min), frequency of awakening at night (times), and duration of napping (min) 1 week prior to the survey.
3) Pregnancy and birth
(1) Basic information: Age, height, BMI during the non-pregnancy period, family members, occupation, smoking, frequency of spontaneous abortion, and previous infertility treatment.
(2) Pregnancy/birth information: Threatened abortion/ premature labor, duration of birth, placental weight, volume of blood loss, type of birth, and amniotic fluid.
(3) Fetal/neonatal information: Fetal distress, small for gestational age (SGA), umbilical cord blood pH, Ap-gar score, body weight, and height.
4. Analytical method
Relationship between sleep-disordered breathing and perinatal outcome in pregnant women
participate; there are no disadvantages related to with-drawal; and personal information is protected. On the other hand, we promised that they would be referred to a sleep-specialized hospital via the hospital if SDB was suspected based on the results of assessment. The pro-tocol of this study was approved by the Ethics Review Board (Description number: 501).
III. Results 1. ODI evaluation
Of the 179 subjects, 3%ODI was less than 0.5 in 38 (21.2%), 0.5 to 4.9 in 119 (66.5%), and 5 to 14.9 in 22 (12.3%) (Table 1). Therefore, we compared a mild SDB group (3%ODI<5, n=22) with a normal group (3%ODI<5, n=157).
2. Subjects' backgrounds
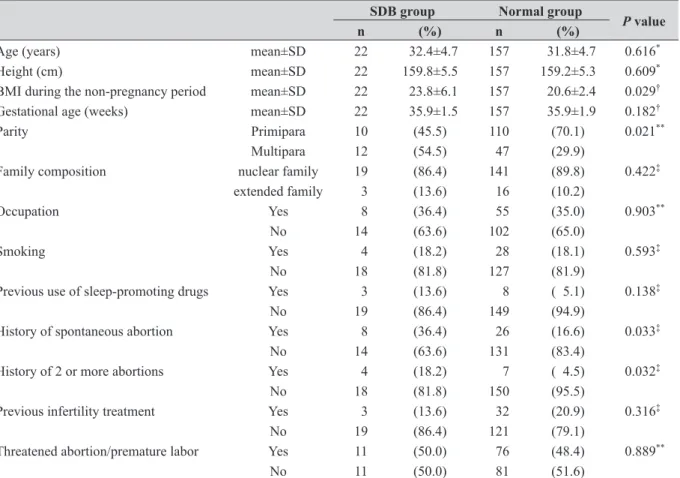
The mean ages in the SDB and normal groups were 32.4±4.7 (mean±SD) and 31.8±4.7 (p=0.616) years, re-spectively. The mean gestational ages were 35.9±1.5 and 35.9±1.9 (p=0.182) weeks, respectively. There were no marked differences in the family composition (p=0.422), smoking (p=0.593), previous infertility treatment (p= 0.316), or incidences of threatened abortion/premature labor (p=0.889) (Table 2). However, in the SDB group, the number of multiparas was larger than in the normal group (p=0.021), and the proportion of those with a his-tory of spontaneous abortion was higher (p=0.033).
3. Association between SDB and sleep
In the SDB and normal groups, the frequency of nocturnal awakening was 2.6±1.7 and 1.9±1.1 times, respectively (p=0.042) (Table 3). There were no differ-ences in the interval from bedtime until sleep induction (p=0.775), the total sleep time (p=0.447), or duration of napping (p=0.863).
Chart Ver.1.1 analytical software. When comparing val-ues between 2 groups with and without SDB, we used the Shapiro-Wilk test employing all variables. When 2 samples showed a normal distribution, the t-test was em-ployed. When there was no normal distribution, Mann-Whitney's U test was used. In addition, we performed logistic regression analysis (forced input method) to cal-culate the odds ratio (OR), regarding SDB as a depen-dent variable, turbid amniotic fluid, fetal distress, SGA, and the type of birth as independent variables, and parity and obesity as adjustment variables. For statistical anal-ysis, we employed Dr. SPSSII for Windows software.
5. Measurement procedures
(1) The purpose and contents of this survey should be explained in writing or verbally during the waiting time before a pregnancy checkup or on fetal heart rate monitoring.
(2) When pregnant women consent to participate in this study, they are enrolled as subjects with their signa-ture on the "informed consent form".
(3) Investigators should explain methods to use a "pulse-oximeter" at home and write/collect the "sleep assess-ment sheet".
(4) The pulse-oximeter should be brought to their home. At the start of sleep at night, it must be attached to the first joint of the second, third, or fourth fingers on the non-handed side. After waking-up, it should be removed, and the time of attachment/removal should be recorded on the sleep assessment sheet. Measure-ment is performed for one evening.
(5) After the completion of measurement, the pulse-ox-imeter should be returned to the hospital by post or on a subsequent pregnancy checkup.
(6) The results of screening with the pulse-oximeter should be directly explained by the investigator, or sent by mail based on their wishes.
6. Ethical consideration
After explaining the following items verbally and in writing, informed consent was obtained from all sub-jects: the purpose of this study; subjects can participate in this study based on their free will; they can with-draw from this study at any time even after agreeing to
Table 1. Frequency of SDB (n=179) evaluation 3%ODI n % none normal mild moderate severe <0.5 0.5~4.9 5.0~14.9 15.0~29.9 ≥30.0 38 119 22 0 0 21.2 66.5 12.3
4. Association between SDB and birth
There were no marked differences in the duration of birth (p=0.102), volume of blood loss (p=0.358), neonatal body weight (p=0.405), umbilical cord blood pH (p=0.128), or Apgar scores at 1 and 5 min (p=0.272 and 0.147, respectively) between the SDB and normal groups (Table 4). The placental weight in the former was slightly lower than in the latter (p=0.067). The pla-cental coefficient (calculated by dividing the plapla-cental
weight by the neonatal body weight) was higher in the former (p=0.032).
The prevalence of emergency CB/ vacuum extrac-tion was 45.4% in SDB and 26.1% in normal group (p=0.060). The fetal state are 4.5% SGA, 23.8% turbid amniotic fluid, 31.8% fetal distress in SDB, and 2.5% SGA, 19.7% turbid amniotic fluid, 24.2% fetal distress in normal group.
P value
n (%) n (%)
Age (years) mean±SD 22 32.4±4.7 157 31.8±4.7 0.616*
Height (cm) mean±SD 22 159.8±5.5 157 159.2±5.3 0.609*
BMI during the non-pregnancy period mean±SD 22 23.8±6.1 157 20.6±2.4 0.029†
Gestational age (weeks) mean±SD 22 35.9±1.5 157 35.9±1.9 0.182†
Parity Primipara 10 (45.5) 110 (70.1) 0.021**
Multipara 12 (54.5) 47 (29.9)
Family composition nuclear family 19 (86.4) 141 (89.8) 0.422‡
extended family 3 (13.6) 16 (10.2)
Occupation Yes 8 (36.4) 55 (35.0) 0.903**
No 14 (63.6) 102 (65.0)
Smoking Yes 4 (18.2) 28 (18.1) 0.593‡
No 18 (81.8) 127 (81.9)
Previous use of sleep-promoting drugs Yes 3 (13.6) 8 ( 5.1) 0.138‡
No 19 (86.4) 149 (94.9)
History of spontaneous abortion Yes 8 (36.4) 26 (16.6) 0.033‡
No 14 (63.6) 131 (83.4)
History of 2 or more abortions Yes 4 (18.2) 7 ( 4.5) 0.032‡
No 18 (81.8) 150 (95.5)
Previous infertility treatment Yes 3 (13.6) 32 (20.9) 0.316‡
No 19 (86.4) 121 (79.1)
Threatened abortion/premature labor Yes 11 (50.0) 76 (48.4) 0.889**
No 11 (50.0) 81 (51.6) SDB: sleep disordered breathing; BMI: body mass index
*: t-test, †: Mann-Whitney’s U test, **: Pearson’s chi-squared test, ‡: Fisher’s exact probability test
Table 3. Sleep in the SDB and normal groups
SDB group Normal group
P value
n Mean Standarddeviation n Mean Standarddeviation
Interval until sleep induction (min) Total sleep time (min)
Frequency of nocturnal awakenings (times) Duration of napping (min)
22 22 22 22 29.8 457.3 2.6 80.5 24.2 94.0 1.7 73.1 155 153 151 153 27.2 449.2 1.9 73.4 22.7 76.9 1.1 50.1 0.775 0.447 0.042 0.863 Mann-Whitney’s U test
Relationship between sleep-disordered breathing and perinatal outcome in pregnant women
5. OR of fetal/birth factors for SDB
We calculated the adjusted OR, regarding parity and obesity as confounding variables (Table 5). The adjusted ORs of turbid amniotic fluid, fetal distress, and SGA were 0.80 (95% confidence interval (CI): 0.43-1.50), 1.64 (95%CI: 0.54-4.95), and 3.15 (95%CI: 0.31-32.13), respectively. On the other hand, regarding
spontaneous vaginal birth as the baseline, the adjusted OR of elective or emergency CB/vacuum extraction was 3.03 (95%CI: 1.10-8.33). Regarding elective CB/spon-taneous vaginal birth as the baseline, the adjusted OR of emergency CB/vacuum extraction was 5.18 (95%CI: 1.44-18.65).
Table 4 Birth and neonatal state in the SDB and normal groups
SDB group Normal group
P value
n Mean Standarddeviation n Mean Standarddeviation
Interval until birth (days) 22 279.4 9.5 157 278.9 8.3 0.819*
Duration of birth (min) 12 429.1 257.1 111 699.4 531.3 0.102†
Type of birth(%)
spontaneous vaginal birth 9 40.9(%) 99 63.1(%) vacuum extraction 3 13.6(%) 12 7.6(%) elective cesarean birth 3 13.6(%) 17 10.8(%) emergency cesarean birth 7 31.8(%) 29 18.5(%)
Placental weight (g) 20 623.2 105.2 155 580.0 98.5 0.067†
Placental coefficient: pl(g)/wt(g) 20 0.205 0.034 155 0.191 0.026 0.032*
Volume of blood loss (g) 22 582.4 343.0 157 517.4 325.0 0.358†
Birth weight (g) 22 3094.3 346.9 157 3035.0 307.4 0.405* Birth height (cm) 22 49.1 1.4 157 48.6 1.6 0.161* Head circumference (cm) 22 33.7 1.2 155 33.2 1.2 0.169† Chest circumference (cm) 22 32.3 1.4 155 31.9 1.3 0.266* Head circumference (cm)/ chest circumference (cm) 22 1.05 0.05 155 1.04 0.04 0.699† Umbilical cord blood pH 21 7.253 0.096 147 7.285 0.066 0.128†
Apgar score at 1 minute (points) 22 8.5 1.0 156 8.7 1.0 0.272†
Apgar score at 5 minutes (points) 22 9.6 0.5 156 9.8 0.5 0.147†
Small for gestational age (%) 1 4.5(%) 4 2.5(%) Turbid amniotic fluid (%) 5 23.8(%) 31 19.7(%) Fetal distress (%) 7 31.8(%) 38 24.2(%) Placental coefficient: placental (g)/birth weight(g) *: t test, †: Mann-Whitney’s U test
Table 5. Association between SDB and the fetal state/type of birth
Adjusted OR 95% Confidence interval
Fetus Turbid amniotic fluid Present 0.80
Absent 1.00 0.43- 1.50 Fetal distress Present 1.64
Absent 1.00 0.54- 4.95
SGA Present 3.15
Absent 1.00 0.31-32.13 Birth Spontaneous vaginal birth 1.00
Elective/emergency CB, vacuum extraction 3.03 1.10- 8.33 Elective CB, Spontaneous vaginal birth 1.00
Emergency CB, vacuum extraction 5.18 1.44-18.65 -The presence or absence of parity and obesity (BMI>25) was adjusted.
In this study, no pregnant woman showed severe/ moderate SDB. However, mild SDB (3%ODI: 5 to 14.9) was observed in 12.3% of the subjects. We employed a criterion to screen for mild SDB, a 3%ODI of 5 episodes or more; treatment was not necessary. However, in the SDB group, the proportion of patients who underwent elective or emergency CB/vacuum extraction was high-er, although there was no association between SDB and turbid amniotic fluid/fetal distress. In pregnant women with SDB, maternal hypoxia related to periodic apnea during sleep may influence the fetal condition and type of birth. Fetal development/growth disorder related to hypoxia may reduce the birth weight. Emergency CB is mainly indicated for patients with fetal distress. A previ-ous study regarding sleep and the type of birth involving pregnant women indicated that a sleep time of 6 hours or less prolonged the duration of labor pains, increasing the rate at which CB was selected (Lee & Gay, 2004). This suggests that a short sleep time affects fetal growth and the expulsive force. Furthermore, another study re-ported that the Apgar score and body weight were low in neonates born to women with obstructive sleep ap-nea (Sahin, Koken, Cosar, et al., 2008). In this study, even patients with mild SDB showed abnormalities in the type of birth; SDB is speculated to have influence on birth. Therefore, mild SDB should be treated in the early stage.
In addition, it has been shown that sleep is not deep in most pregnant women in the third trimester (Horiu-chi, Kondo, Koyama, et al.,1990 ; Lee, Zaffke, Mc En-any, 2000). Therefore, it is recommended that a sleep time of 8 hours or more should be maintained in this phase (Lee & Gay, 2004). In this study, the mean sleep time in the SDB group was 7.5 hours or more. Not only the duration of sleep but also its quality is important; the two factors should be reviewed.
To evaluate SDB in pregnant women, one study employed a questionnaire survey (Pien, Fife, Pack, et al., 2005), and another study performed polysomnog-raphy after selecting subjects via a questionnaire sur-vey using the two-stage method (Sahin, Koken, Cosar, et al ., 2008). Polysomnography is a gold standard for
the examination fee is expensive; cost-performance for diagnosis is low. On the other hand, the pulse-oximeter employed in this study facilitates measurement at home, and is inexpensive. In addition, its usefulness in SDB assessment in comparison with polysomnography has been demonstrated (Nakamata, Kubota, Sakaki, 2003). Considering these merits, SDB assessment with a pulse-oximeter should be further applied in clinical practice.
However, the limitations of this study include the fact that it employed a one-point survey system involv-ing the third trimester without continuous follow-up. Among pregnant women who were managed in one lo-cal hospital, the subjects were those who consented to participate in this study; this survey did not involve all pregnant women. Measurement with a pulse oximeter was performed on only one evening. Activities may have influenced their sleep on the day of measurement. In addition, the appropriate timing, frequency, and pe-riod of SDB screening during pregnancy should be in-vestigated in the future.
V. Conclusion
Mild SDB was observed in 12.3% of the third-trimester pregnant women. There was no association between SDB and fetal distress/turbid amniotic fluid. However, SDB was associated with the type of birth. The results of this study suggest that even mild SDB influences the perinatal outcome in pregnant women. Therefore, screening with a pulse-oximeter should be effectively utilized in multiparas with a history of spon-taneous abortion and pregnant women complaining of frequent awakening at night or sleep disturbance. In ad-dition, early intervention to improve the state of sleep and treat sleep disturbance may be necessary to promote a safer pregnancy.
References
Horiuchi, C., Kondo, J., Koyama, M., Kido, H., Ohkubo, N., & Yamamoto, T., et al (1990). Nocturnal Sleep Dur-ing Pregnancy and Postpartum Periods The Relation-ship Between Subjective Evaluation and Polygrapphic
Relationship between sleep-disordered breathing and perinatal outcome in pregnant women
Findings-.J. Jpn. Acad. Nurs. Sci, 10 (2), 8-17. Inoue, Y. & Yamashiro, Y. (2006). Sleep disordered
breath-ing Update 2006. Tokyo.
Lee, K.A. & Gay, C.L. (2004). Sleep in late pregnancy pre-dicts length of labor and type of delivery. Am J Obstet Gynecol, 191, 2041-2046.
Lee, K.A., Zaffke, M.E. & McEnany, G. (2000). Parity and sleep patterns during and after pregnancy. Obstet Gy-necol, 95(1), 14-18.
Mindell, J.A. & Jacobson, B.J. (2000). Sleep disturbances during pregnancy. J Obstet gynecol Neonatal Nurs, 29(6), 590-597.
Nakamata, M., Kubota, Y., Sakai, K., Kinefuchi, S., Nakaya-ma, H., & Ohdaira, T., et al (2003). The limitation of screening test for patients with sleep apnea syndrome using pulse oximetry. J Jpn Soc respire Care, 12(3), 401-406.
Pien, G.W., Fife, D., Pack. A.I., Nkwuo, J.E., & Schwab, R.J.(2005). Changes in symptoms of sleep-disordered breathing during pregnancy. Sleep, 28(10), 1299-1305. Sahin, F.K., Koken, G., Cosar, E., Saylan, F., Fidan, F., &
Yilmazer, M., et al. (2008). Obstructive sleep apnea in pregnancy and fetal outcome. Int J Gynecol Obstet, 100(2),141-146.
Shinkawa, H., Shimada. M. & Fujita, T. (2008). Sleep qual-ity and sleepiness characteristics in first trimester ex-pectant mothers. J. Japan. Acad. Midwif, 22(2), 180-188.
Shiomi. T. & Yabushita. H. (2006).The Japanese journal for midewife. 60 (9), 816-820.
Shiomi. T., Yabushita. H. & Wakastuki. A. (2005). Japaanese medical journal, 4332, 33-36.
Sleep Disorder meeting (2005). Sleep apnea syndromes- Practice Guideline. Tokyo.
Suzuki, K., Ohida, T., Sone. T., Yokoyama, E., Miyake, T., & Harano, S., et al. (2003). An Epidemiology Study of Sleep Problems Among the Japanese Pregnant Women. Jpn J publish Health, 50 (6), 526-539.
Young, T., Palta, M., Dempsey, J., Skatrud, J., Weber, S., & Badr, S. (1993). The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med, 328(17), 1230-1235.
宮 川 幸 代*1,江 守 陽 子*1,川 野 亜津子*1 櫻 井 進*2,谷 川 武*2 *1筑波大学大学院人間総合科学研究科 *2愛媛大学大学院医学系研究科 抄 録 目 的
本研究の目的は,妊婦の睡眠呼吸障害(Sleep Disordered Breathing: SDB)が出産のアウトカムに及ぼ す影響を検討することにある。
方 法
妊婦健康診査を受けている妊娠28週以降の妊婦179名を対象とし,パルスオキシメータにより,夜 間睡眠中の睡眠1時間あたりの末梢動脈血中の酸素飽和度低下指数(oxygen desaturation index: ODI)を 算出した。判定基準はSDBなし(3%ODI<0.5),正常範囲内(0.5 3%ODI<5),軽度(5 3%ODI<15), 中等度(15 3%ODI<30),重度(3%ODI 30)の5区分とした。分析は,多重ロジスティック回帰分析に よって,SDBと出産のアウトカムとの関連を検討した。
結 果
ODI判定は3%ODI<0.5は38名(21.2%),0.5 3%ODI<5は119名(66.5%),5 3%ODI<15は22名(12.3%), 15 3%ODIは0名であった。したがって,軽度SDB群(3%ODI 5)22名と正常群(3%ODI<5)157名の2 群間の比較を行なった。出産歴および肥満を交絡因子とした調整オッズ比は,「自然出産」を基準とする と「予定or緊急帝王切開出産Cesarean Birth(CB)・吸引出産」での調整ORは3.03(95%CI: 1.10-8.33),「予 定CB・自然出産」を基準とすると「緊急CB・吸引出産」での調整ORは5.18(95%CI: 1.44-18.65)であった。 結 論 妊娠女性におけるSDBは軽度でも出産のアウトカムに問題となることが示唆されたことから,自然 流産歴を持つ経産婦,夜間の覚醒回数や睡眠問題について訴えの多い妊婦に対しては,パルスオキシ メータによるスクリーニングを有効に活用するとともに,より安全な出産ケアの提供のためには速やか な睡眠状況の改善や睡眠問題の治療のための介入が必要と思われる。 キーワード:睡眠呼吸障害,妊婦,パルスオキシメータ,スクリーニング