1.
3
2.
3
3.
4
4.
5
5.
10
6.
12
7.
14
8.
14
9.
14
10.
17
cancer stem-like cells: CSLCs CSLCs CSLCs(Pancreatic CSLCs: P-CSLCs) P-CSLCs 2 P-CSLCs P-CSLCs n=80 Cox P-CSLCs Calreticulin (CRT) CRT P-CSLCs P-CSLCs CD44v9
CRThigh/CD44v9low ATP binding cassette
CRTlow/CD44v9high CRT CRT P-CSLCs CRT P-CSLCs P-CSLCs CRT P-CSLCs 4 2014 15-20% 1
cancer
stem-like cells (CSLCs) 2-4
CSLCs
pancreatic CSLCs P-CSLCs CD24 CD44 epithelial-specific antigen (ESA)
5 CD133 6 aldehyde dehydrogenase 1
(ALDH1) 7 c-Met 8 doublecortin-like kinase 1 (DCLK1) 9 P-CSLCs
CSLCs CD44 variant isoform CD44v9
CD44v9 reactive oxygen species
(ROS) 10 P-CSLCs 11 CD24 CD44 ESA CD44v9 P-CSLCs CD44v9 P-CSLCs 10, 12 P-CSLCs Calreticulin (CRT) 46–65-kDa Ca2+ HLA class mitoxantrone oxaliplatin CRT 13, 14 CRT 15 CRT P-CSLCs Proteomics P-CSLCs
4-1
YPK2 YPK516 (Life Technologies,
Tokyo, Japan) 10% DMEM-F12 (Sigma-Aldrich Japan, Tokyo, Japan) 37 °C 5%
CO2
4-2 P-CSLCs
LIF (Merck Millipore, Darmstadt, Germany) NSF-1 (Lonza, Tokyo, Japan) N-acetyl-L-cysteine
(NAC; Sigma-Aldrich) 1 sphere
sphere B27 supplement (Life Technologies) EGF
(Sigma-Aldrich Japan) bFGF (Merck Millipore)
1 1-2
YPK2-Lm YPK5-Lm
Proteomics P-CSLCs
4-3 2
Dead Cell Removal MicroBeads (Miltenyi Biotec, Gladbach, Germany) MidiMACS™
Separator (Miltenyi Biotec) CD44v9 rat IgG (clone RV3, Cosmo bio, Tokyo, Japan)
anti-rat mouse IgG (eBioscience, San Diego, CA, USA) microbeads anti-biotin mouse
IgG (Miltenyi Biotec) MidiMACS™ Separator CD44v9
YPK YPK-Lm CD44v9 0.2% pharmalyte
5M Urea, 2M Thiourea, 2% CHAPS, 2% SB3-10, 1% DTT
0.34ml 18 cm Immobiline Drystrip(pH 3–10, GE Healthcare,
Electrophoresis Unit CoolPhoreStar IPG-IEF Type-P (Anatech, Tokyo, Japan)
500 V 1 3,500 V 7.5 A 50 mM Tris-HCl
(pH 6.8), 6 M urea, 32% glycerol, 10% SDS, 0.25% DTT B 50 mM Tris-HCl
(pH 6.8), 6 M urea, 32% glycerol, 10% SDS, 4.5% , 0.125%
Immobiline Drystrip (9–18% acrylamide; Towa
Environment Science, Osaka, Japan) ANDERSON ISO-DALT Multiple Electrophoresis System
(Hoefer, Holliston, MA, USA) 80 V 16 SDS
SYPRO Ruby protein gel stain (S21900;
Thermo Fisher Scientific, Waltham, MA, USA) Molecular Imager FX
(Bio-Rad, Tokyo, Japan) ImageMaster 2D Platinum software (GE Healthcare)
YPK2-Lm YPK5-Lm 4-4 100 mM 20 50 mM 5 mM 0.01 g/ l 37 16 5% TFA 5%TFA 50% 20 3 10 l
ZipTip C18 pipette tips (ZTC18S960, Merck Millipore)
50% 0.1%TFA 1 l
(0.3 g/l alpha-cyano-4-hydroxycinnamic acid, 33% acetone, 66% ethanol target plate
(MTP Anchorchip 600/384, Bruker Daltonics, Bremen, Germany) (Ultraflex
TOF/TOF; Bruker Daltonics) MS/MS NCBInr
Mascot database search engine (Matrix Science, London, UK) 4-5
50ml 2% PBS 2 × 105 cells/100 l
1
1 isotype 20 4
1
rat anti-CD44v9 (clone RV3, Cosmo bio, Tokyo, Japan)
mouse Alexa Fluor 488 anti-CRT (clone #326203, R&D systems, Minneapolis, MN, USA) Vioblue anti-CD47 (#130-101-359, Miltenyi Biotec, Gladbach, Germany)
CD44v9 2 allophycocyanin donkey anti-rat IgG (eBioscience)
2 CRT
CD44v9 Fix/Permeabilization buffer (eBioscience) 20
PBS permeabilization buffer (eBioscience) 10
Blocking buffer (2% normal rat serum in permeabilization buffer) Alexa Fluor 488 anti-CRT (clone 326203, R&D systems)
isotype control 20 Permeabilization buffer 2% FBS
PBS MACSQuant analyzer (Miltenyi Biotec)
Relative fluorescence intensity (RFI)
= [mean fluorescence intensity (MFI) - MFI of corresponding isotype control]/MFI of corresponding isotype control17
4-6
YPK-Lm CRT, CD44v9 BD FACSAriaII (BD Biosciences, San Jose, CA,
USA) CRThigh/CD44v9low CRTlow/CD44v9high CRThigh/CD44v9high 3
4-7 ATP ABC
ABC 18
side population (SP) ABC Hoechst 33342
19 ABC YPK2-Lm YPL5-Lm
5% FBS DMEM 5 g/ml Hoechst 33342 (Sigma-Aldrich Japan) 30 min
BD LSRFortessa X-20 cell analyzer (BD Biosciences)
Hoechst 33342 375-nm trigon violet laser 450/20 (Hoechst 33342-Blue) 670 LP
(Hoechst 33342-Red) 4-8 (1) 2001 6 2013 6 2007 3 2012 10 D2 20 IPMN ( H27-007) (2)
10 mM Target Retrieval Solution, pH 6.0 (Dako, Tokyo, Japan)
CRT 3% H2O2 10 CD44v9 0.3% H2O2
5 Protein block serum-free (Dako)
1 2
1 mouse anti-CRT monoclonal antibody (clone FMC75, Abcam,
Cambridge, MA, USA): 1:6000 1
30
1 rat anti- CD44v9 monoclonal antibody (clone RV3, Cosmo bio)
1:200,4 °C, overnight
2 HRP anti-rat IgG (Nichirei Biosciences, Tokyo, Japan).
PBS 3,3 -diaminobenzidine tetrahydrochloride (DAB; Dako) 3
21
a) absent / weak :1 b) moderate: 2 c) strong: 3
IHC × % (=1×absent / weak % 2×moderate %
3×strong % )
(3)
citrate buffer (10 mM, pH 6.0) 95 °C 10 5% 0.3% Triton
X-100 PBS 60 CRT CD44v9
Anti-CRT antibody (FMC75, Abcam) 1:200
1 Alexa Fluor 488 anti-mouse IgG (#4408, Cell Signaling Technology, Denver, MA, USA)
1:1,000 1.5 anti-CD44v9 rat antibody (RV3, Cosmo bio) 1:100 1
Alexa Fluor 555 anti-rat IgG (#4417, Cell Signaling Technology) 1:1,000 1.5
DAPI ProLong Gold Antifade Reagent (#8961; Cell Signaling Technology)
4-9
± Student’s t-test Chi-squared
Kaplan-Meier Wilcoxon test
Japan) p 0.05
5-1. Calreticulin
YPK2-Lm 4.43 YPK5-Lm 5.80
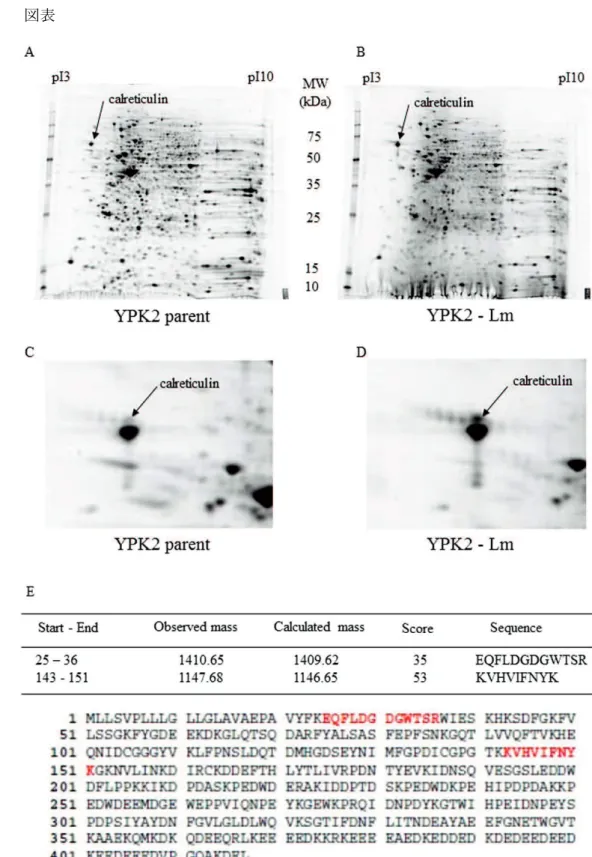
(Fig. 1A–D, ) Calreticulin (CRT) (NCBI accession No. gi|4757900) (Fig. 1E)
5-2. YPK-Lm CRT CD44v9
YPK2-Lm YPK5-Lm
CRT CD44v9 (Fig. 2A, B) YPK-Lm CRThigh/CD44v9low
CRTlow/CD44v9high 2 (Fig. 2C, D) CRT YPK-Lm
(Fig. 2E, F)
5-3.YPK-Lm CD47
CD47 CRT ”eat me signal” “anti-phagocytic signal” 22
CD47 YPK-Lm
Fig. 3A 22 CRT CD47
Fig. 3B
5-4. YPK-Lm ABC
YPK2 side population ( SP) 0.338% YPK2- Lm
34.0% (Fig. 2G) YPK5-Lm SP 12.9%
SP(1.72%) (Fig. 2H) YPK- Lm CRThigh/CD44v9low SP
(Fig. 4B, F) CRTlow/CD44v9high (Fig. 4C, G) CRThigh/CD44v9high (Fig. 4D, H)
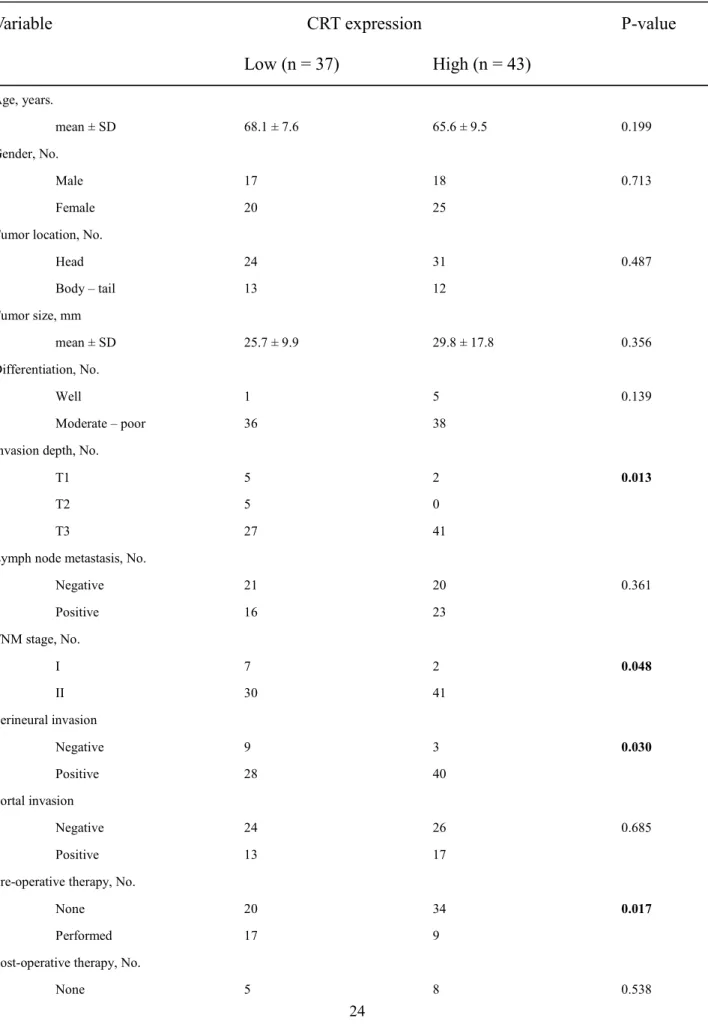
subpopulation 5-5 CRT CD44v9 n=77 n=64 61 80 Fig. 5 1/3 (n = 26) (n = 67) CRT Fig. 6A CRT CD44v9 Fig. 6B CD44v9 CRT CD44v9 IHC ( : 0.356 [0.148–0.534], p = 0.0012) Cox 23-25 TNM T CRT CD44v9 IHC CRT IHC (p < 0.01) (p < 0.01) (p < 0.05) (Table 1) 1 CRT IHC 150 CRT
(IHC score 150; n = 43) CRT (IHC score < 150; n = 37)
CD44v9 (IHC score 165; n = 40) CD44v9 (IHC score < 165; n = 40)
CRT Table 2 CRT
T3 Stage II CD44v9
CRT (p =
CRT 26 CRT IHC CRT 4-7 CRT 5-5 CRT CD44v9 CRT CD44v9 CRT CD44v9 (Fig. 6C) CRT P-CSLCs CRT CRT ER Ca2+ folding CRT ER 27 CRT ER fold
CRT ”unfolded protein response”
26 CRT CD44v9 (Fig. 6C) CRT CD44v9 population 2 1 P-CSLCs P-CSLCs CRT CRT SP CSLCs 28 SP
29 CRThigh/CD44v9low CRTlow/CD44v9high SP CRT CD44v9 P-CSLCs CRT CSLCs 22 CSLCs CRT CRT Chao 22 CRT CRT CRT “eat-me” signal 13 CRT 30 31 15 2 22 1 CRT anti-phagocytic signal CD47 CRT CD47 CD47 YPK-Lm P-CSLCs CD47 2 CRT CRT neuropilin-1, matrix
metalloproteinase (MMP)2, MMP9, focal adhesion kinase (FAK) upregulate
32 phosphoinositide 3-kinase (PI3K)/Akt pathway
anoikis
33 CRT
CRT LDL-receptor related protein 1
(LRP1)/CD91 34
tumor-associated macrophages (TAMs) M2
35 CRT
36 CRT CRT P-CSLCs CRT P-CSLCs CD24, CD44, ESA UV P-CSLCs CRT
[1] Ryan DP, Hong TS, Bardeesy N. Pancreatic adenocarcinoma. N Engl J Med. 2014; 371: 1039-49. [2] Clevers H. The cancer stem cell: premises, promises and challenges. Nat Med. 2011; 17: 313-9.
[3] Li X, Lewis MT, Huang J, et al. Intrinsic resistance of tumorigenic breast cancer cells to chemotherapy. J Natl Cancer Inst. 2008; 100: 672-9.
[4] Zhou BB, Zhang H, Damelin M, Geles KG, Grindley JC, Dirks PB. Tumour-initiating cells: challenges and opportunities for anticancer drug discovery. Nat Rev Drug Discov. 2009; 8: 806-23.
[5] Li C, Heidt DG, Dalerba P, et al. Identification of pancreatic cancer stem cells. Cancer research. 2007; 67: 1030-7.
[6] Hermann PC, Herrler T, Aicher A, et al. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2007; 1: 313-23.
[7] Rasheed ZA, Yang J, Wang Q, et al. Prognostic significance of tumorigenic cells with mesenchymal features in pancreatic adenocarcinoma. J Natl Cancer Inst. 2010; 102: 340-51.
[8] Li C, Hynes M, Dosch J, et al. c-Met is a marker of pancreatic cancer stem cells and therapeutic target. Gastroenterology. 2011; 141: 2218-27.
[9] Sureban SM, May R, Qu D, et al. DCLK1 regulates pluripotency and angiogenic factors via microRNA-dependent mechanisms in pancreatic cancer. PLoS ONE. 2013; 8: e73940.
[10] Ishimoto T, Nagano O, Yae T, et al. CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc(-) and thereby promotes tumor growth. Cancer cell. 2011; 19: 387-400.
[11] Watanabe Y, Yoshimura K, Yoshikawa K, et al. A stem cell medium containing neural stimulating factor induces a pancreatic cancer stem-like cell-enriched population. Int J Oncol. 2014; 45: 1857-66.
[12] Yoshida GJ, Saya H. Therapeutic strategies targeting cancer stem cells. Cancer Sci. 2016; 107: 5-11. [13] Obeid M, Tesniere A, Ghiringhelli F, et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med. 2007; 13: 54-61.
[14] Yamamura Y, Tsuchikawa T, Miyauchi K, et al. The key role of calreticulin in immunomodulation induced by chemotherapeutic agents. Int J Clin Oncol. 2015; 20: 386-94.
[15] Sheng W, Chen C, Dong M, et al. Overexpression of calreticulin contributes to the development and progression of pancreatic cancer. J Cell Physiol. 2014; 229: 887-97.
[16] Yamamoto K, Yahara N, Gondo T, Ishihara T, Oka M. Establishment and characterization of a new human pancreatic cancer cell line, YPK-1. Bull Yamaguchi Med Sch. 2002; 49: 33-42.
[17] Soga F, Katoh N, Inoue T, Kishimoto S. Serotonin activates human monocytes and prevents apoptosis. J Invest Dermatol 2007;127:1947-55.
[18] Zhou J, Wang CY, Liu T, et al. Persistence of side population cells with high drug efflux capacity in pancreatic cancer. World J Gastroenterol. 2008; 14: 925-30.
[19] Goodell MA, Brose K, Paradis G, Conner AS, Mulligan RC. Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J Exp Med. 1996; 183: 1797 - 806.
[20] Japan Pancreas Society. General Rules for the Study of Pancreatic Cancer, The 6th Edition, Revised Version edn. Tokyo, Japan: Kanehara, 2013
[21] Lee HJ, Xu X, Choe G, et al. Protein overexpression and gene amplification of epidermal growth factor receptor in nonsmall cell lung carcinomas: Comparison of four commercially available antibodies by immunohistochemistry and fluorescence in situ hybridization study. Lung Cancer. 2010; 68: 375-82.
[22] Chao MP, Jaiswal S, Weissman-Tsukamoto R, et al. Calreticulin is the dominant pro-phagocytic signal on multiple human cancers and is counterbalanced by CD47. Sci Transl Med. 2010; 22: 2: 63-84.
[23] Chatterjee D, Katz MH, Rashid A, et al. Perineural and intraneural invasion in posttherapy pancreaticoduodenectomy specimens predicts poor prognosis in patients with pancreatic ductal adenocarcinoma. Am J Surg Pathol. 2012; 36: 409-17.
[24] Lim JE, W. CM, Earle CC. Prognostic factors following curative resection for pancreatic adenocarcinoma: a population-based, linked database analysis of 396 patients. Ann Surg. 2003; 237: 74-85.
[25] Richter A, Niedergethmann M, Sturm JW, Lorenz D, Post S, Trede M. Long-term results of partial pancreaticoduodenectomy for ductal adenocarcinoma of the pancreatic head: 25-year experience. World J Surg. 2003; 27: 324-9.
[26] Panaretakis T, Kepp O, Brockmeier U, et al. Mechanisms of pre-apoptotic calreticulin exposure in immunogenic cell death. The EMBO Journal. 2009; 28: 578-90.
[27] Ye R, Mareninova OA, Barron E, et al. Grp78 heterozygosity regulates chaperone balance in exocrine pancreas with differential response to cerulein-induced acute pancreatitis. Am J Pathol. 2010; 177: 2827 - 36.
[28] Clarke MF, Dick JE, Dirks PB, et al. Cancer stem cells--perspectives on current status and future directions: AACR Workshop on cancer stem cells. Cancer research. 2006; 66: 9339 - 44.
[29] Li D, Su D, Xue L, Liu Y, Pang W. Establishment of pancreatic cancer stem cells by flow cytometry and their biological characteristics. Int J Clin Exp Pathol. 2015; 8: 11218 - 23.
[30] Du XL, Lin DE, Xia SH, et al. Proteomic profiling of proteins dysregulted in Chinese esophageal squamous cell carcinoma. J Mol Med. 2007; 85: 863-75.
[31] Chen CN, Chang CC, Su TE, et al. Identification of calreticulin as a prognosis marker and angiogenic regulator in human gastric cancer. Ann Surg Oncol. 2009; 16: 525-33.
[32] Shi F, Shang L, Pan BQ, et al. Calreticulin promotes migration and invasion of esophageal cancer cells by upregulating neuropilin-1 expression via STAT5A. Clin Cancer Res. 2014; 20: 6153-62.
[33] Du XL, Yang H, Liu SG, et al. Calreticulin promotes cell motility and enhances resistance to anoikis through STAT3-CTTN-Akt pathway in esophageal squamous cell carcinoma. Oncogene. 2009; 28: 3714-22. [34] Gardai SJ, McPhillips KA, Frasch SC, et al. Cell-surface calreticulin initiates clearance of viable or apoptotic cells through trans-activation of LRP on the phagocyte. Cell. 2005; 123: 321-34.
[35] Solinas G, Germano G, Mantovani A, Allavena P. Tumor-associated macrophages (TAM) as major players of the cancer-related inflammation. J Leukoc Biol. 2009; 86: 1065-73.
[36] Bruttel VS, Wischhusen J. Cancer stem cell immunology: key to understanding tumorigenesis and tumor immune escape? Front Immunol. 2014; 29: 360.
Fig. 1. Calreticulin
A-D: YPK2 A, C YPK2-Lm B, D 2
Fig. 2.
A, B: (A)YPK2 YPK2-Lm (B) YPK5 YPK5-Lm CRT
CD44v9
CRT CD4v9
E, F: (E)YPK2 YPK2-Lm F (E)YPK5 YPK5-Lm
CRT
G,H: (G)YPK2 YPK2-Lm H (E)YPK5 YPK5-Lm
Hoechst33342
Fig.3. CD47
A: YPK YPK-Lm CD47
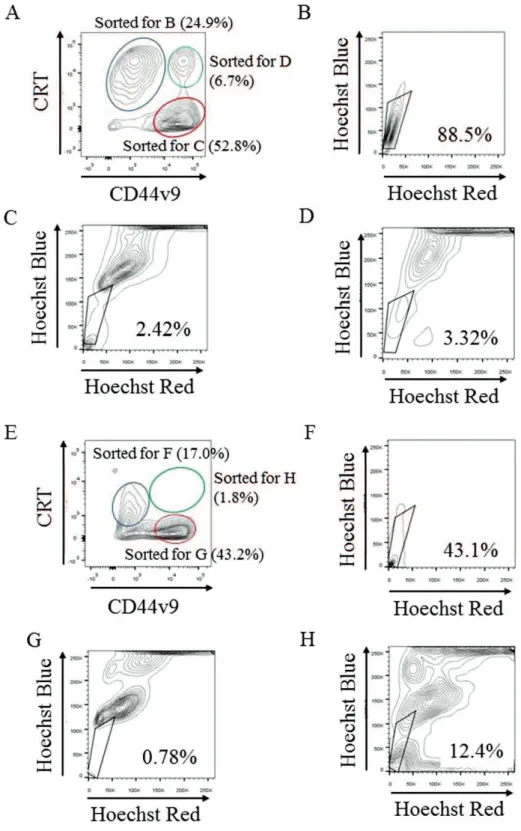
Fig. 4. YPK-Lm sort Side population (SP)
(A) YPK2-Lm calreticulin (CRT)high/CD44 variant isoform 9 (CD44v9)low ( ) CRTlow/CD44v9high
( ) CRThigh/CD44v9high ( ) 3 sort ATP-binding cassette transporter
(D) CRThigh/CD44v9high SP 3.32% (E) YPK5-Lm CRThigh/CD44v9low
( ) CRTlow/CD44v9high ( ) CRThigh/CD44v9high ( ) sort ATP-binding cassette transporter
(F) CRThigh/CD44v9low SP 43.1% (G) CRTlow/CD44v9high SP
0.78% (H) CRThigh/CD44v9high SP 12.4%
Fig.5.
IHC; immunohistochemistry,
A B C Fig.6: Calreticulin (CRT) CD44v9 islet acinus duct
Normal tissue Absent Weak
Strong Moderate HE CRT CD44v9 Merged Merged-DAPI islet acinus duct
Normal tissue Absent Weak
Strong Moderate
A: CRT CRT CRT 50 m B: CD44v9 CD44v9 CD44v9 50 m C: CRT CD44v9 50 m Table 1. Cox
Variable SE p-value Hazard ratio (95% CI)
Age 0.051 0.017 0.002 1.053 (1.019-1.088)
CRT IHC score 0.007 0.002 0.004 1.007 (1.002-1.011)
Post-operative therapy -0.815 0.365 0.026 0.443 (0.216-0.905)
Table 2. CRT
Variable
CRT expression
P-value
Low (n = 37)
High (n = 43)
Age, years. mean ± SD 68.1 ± 7.6 65.6 ± 9.5 0.199 Gender, No. Male 17 18 0.713 Female 20 25Tumor location, No.
Head 24 31 0.487 Body – tail 13 12 Tumor size, mm mean ± SD 25.7 ± 9.9 29.8 ± 17.8 0.356 Differentiation, No. Well 1 5 0.139 Moderate – poor 36 38
Invasion depth, No.
T1 5 2 0.013
T2 5 0
T3 27 41
Lymph node metastasis, No.
Negative 21 20 0.361 Positive 16 23 TNM stage, No. I 7 2 0.048 II 30 41 Perineural invasion Negative 9 3 0.030 Positive 28 40 Portal invasion Negative 24 26 0.685 Positive 13 17
Pre-operative therapy, No.
None 20 34 0.017
Performed 17 9
Post-operative therapy, No.
Performed 32 35 CD44v9 expression, No.
Low 25 15 0.004