9
The Bulletin of Institute of Technologists, No. 8
Article
*1 *2 *2 *3 *4 *1 *2 *3 *4A Study on Test Method for Penetration Depth of Chloride Ions
into Hardened Concrete by Spraying AgNO
3Solution
Kokoro TOBE
*1, Takehiro SAWAMOTO
*2, Kazumasa MORIHAMA
*2,
Yusuke AOKI
*3, and Koji KAWAMATA
*4*1
Undergraduate,
Dept. of Building Technologists, Institute of Technologists
*2Dept. of Building Technologists, Institute of Technologists
*3Dept. of Civil and Environmental Eng.,N.I.T.Kisarazu College
*4
Chuken Consultant Co.,Ltd
Abstract The test method for penetration depth of chloride ions into hardened concrete by spraying AgNO3
solution has been applied since 1970. This test method makes use of discolored boundary between AgCl and Ag2O by spraying AgNO3 solution. However, this method has some problems. In this study,
some problems of this test method were resolved. The main conclusions are as follows. (1) The movement of discolored boundary ends passage of an hour after spraying AgNO3 solution. (2) The
content of total chloride ions at the discolored boundary is about from 2kg/m3 to 4kg/m3. (3) Spraying
NaOH solution before spraying AgNO3 solution is effective when the specimen is carbonated.
Key Words : Concrete, AgNO
3solution, Content of chloride ion, Penetration depth, Discolored Boundary
10
Fig. 1
A
B
B
Fig. 2
Table 1
100 200mm
NaCl
10%
1
0.1mol/L
Photo. 2
15mm
10
20
Fig. 1 Conditions of concrete specimens
Atmosphere
Tide mark
Under the sea
Salt water
Cylinder specimen Core specimen Core specimen Cylinder specimenH.W.L
L.W.L
Photo. 1 Spray AgNO3 Solution to concrete core
Fig. 2 Test flow of this study
Chloride test Carbonation test
Split specimen
Spray distilled water Spray NaOH solution
Spray AgNO3solution
Leave specimen for 60 minutes Dry specimen with hair drier
Measurement for depth of chloride ions Measurement for depth of carbonation Spray C20OH14O4 solution
Wet condition Air-dry condition
11
The Bulletin of Institute of Technologists, No. 8
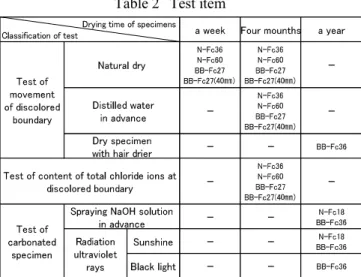
Table 2
1
A
4
B
15
30
1
2
4
Photo. 2 Spray AgNO3 Solution
Photo. 5 Radiate ultraviolet rays Photo. 3 Spray distilled water
Photo. 4 SprayNaOH solution
12
4
Photo. 3
1
4
3.2
(
)
NDIS 3433
7),8),9)Photo. 4
1mol/L
2mol/L
6%
10)Photo. 5
Fig.
3
Fig. 4
Photo. 6
60
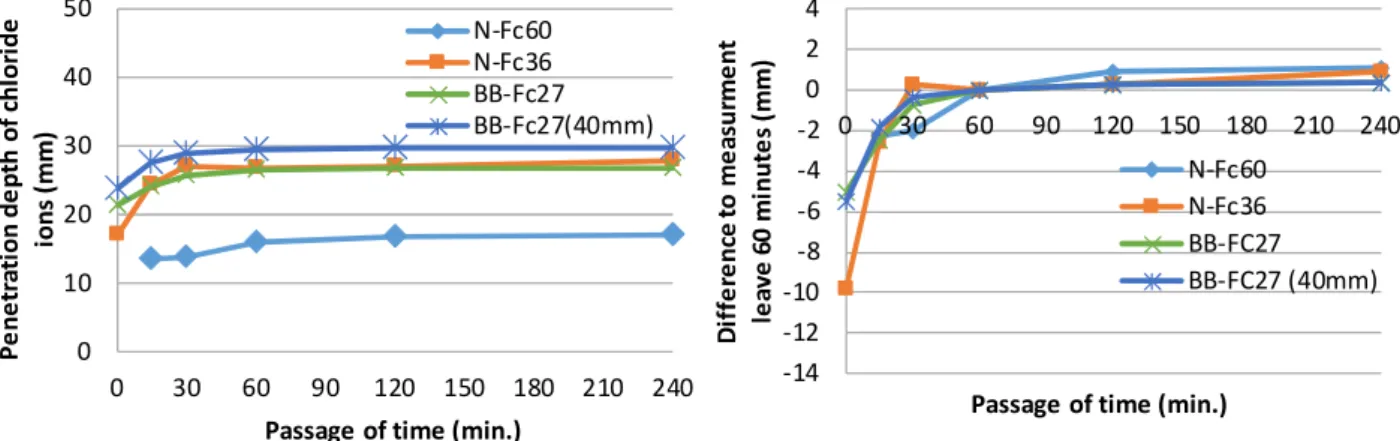
Fig. 3
Fig. 4
60
10mm
Fig. 3
1
Fig. 4
4
N-Fc60
30
Fig. 4
Fig. 3
Fig. 4
Fig. 5
Fig. 5
N-Fc60
15
Photo.
7
Fig. 4
Fig. 5
Fig. 6
Photo. 8
13
The Bulletin of Institute of Technologists, No. 8
Fig. 3 Specimens dried for a week after taking out from salt 0 10 20 30 40 50 0 30 60 90 120 150 180 210 240 Passage of time min.
N-Fc60 N-Fc36 BB-Fc27 BB-Fc27 (40mm) -14 -12 -10 -8 -6 -4 -2 0 2 4 0 30 60 90 120 150 180 210 240
passage of time min. N-Fc60 N-Fc36 BB-FC27 BB-FC27 (40mm)
Fig. 4 Specimens dried for 4 months after taking out from salt water 0 10 20 30 40 50 0 30 60 90 120 150 180 210 240 Passage of time (min.)
N-Fc60 N-Fc36 BB-Fc27 BB-Fc27(40mm) -14 -12 -10 -8 -6 -4 -2 0 2 4 0 30 60 90 120 150 180 210 240
14
15
Fig. 7
Fig. 7
Fig. 5 Specimens dried for 4 months after taking out from salt water Spray distilled water in advance 0 10 20 30 40 50 0 30 60 90 120 150 180 210 240 Passage of time (min.)
N-Fc60 N-Fc36 BB-Fc27 BB-Fc27(40mm) -14 -12 -10 -8 -6 -4 -2 0 2 4 0 30 60 90 120 150 180 210 240
Passage of time (min.) N-Fc60 N-Fc36 BB-FC27 BB-FC27 (40mm)
Fig. 6 Effect of drying specimen with hair drier
0 2 4 6 8 10 12 0 15 30 45 60
Passage of time min.
Natural dry Dry with hair drier
Fig. 7 Relation between content of total chloride ions and distance from concrete surface
Spray AgNO3 solution
Photo. 7 Effect of spraying distilled water in advance N Fc60 Spray distilled water in advance
15
The Bulletin of Institute of Technologists, No. 8
Table 3
60
2kg/m
3B
3kg/m
3Table 3 Content of total chloride ions at discolored boundary
Photo. 10 Spray NaOH solution to carbonated specimen N Fc18
Spray AgNO3 solution Spray NaOH solution
Photo. 9 Spray AgNO3 solution and C20H14O4 solution to
carbonated specimen N Fc18
Spray C20H14O4 solution Spray AgNO3 solution
Spray distilled water in advance
Photo. 11 Effect of spraying NaOH solution in advance BB Fc36 Spray 1mol/L NaOH solution
in advance
Spray distilled water in advance
16
60
2kg/m
360
Photo. 9
Photo.
10
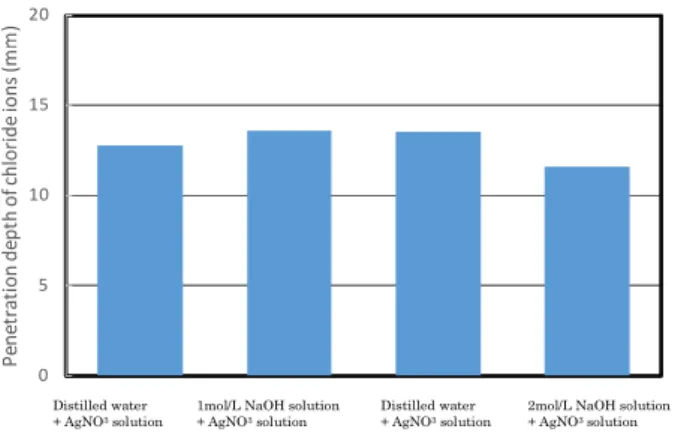
Fig. 8
Photo. 11
1mol/L
2mol/L
Photo. 12
Photo. 13
Photo. 12
10
Discolored boundary by chloride ionsPhoto. 12 Before radiation ultraviolet rays by sunshine Discolored boundary by carbonation
Photo. 14 Radiate ultraviolet rays by black light Photo. 13 After radiation ultraviolet rays by sunshine Discolored boundary
by chloride ions
Discolored boundary by carbonation
Distilled water
+ AgNO3 solution 1mol/L NaOH solution + AgNO3 solution Distilled water + AgNO3 solution 2mol/L NaOH solution + AgNO3 solution
Fig. 8 Effect of spraying NaOH solution in advance BB Fc36 0
17
The Bulletin of Institute of Technologists, No. 8
Photo.
14
10
(1)
60
(2)
60
(3)
2 4kg/m
3(4)
(5)
( ) NDIS 3437 WG1) Nobuaki Otsuki, Shigeyoshi Nagataki, Kenji Nakashita: Evaluation of AgNO3 Solution Spray Method for
Measurement of Chloride Penetration into Hardened Cementitious Matrix Materials, ACI Material Journal / November-December, Title no.89-M64, pp.587-592, 1992 2)
No.42 pp.11-18 1990.12
3) Fuqiang He, Caijun Shi, Qiang Yuan, Changping Chen, Keren Zheng : AgNO3-based colorimetric methods for
measurement of chloride penetration in concrete Construction and Building Materials, Volume 26, Issue 1, January,pp.1-8, 2012.1
4) M. Collepardi : Quick method to determine free and bound chlorides in concrete, RILEM International Workshop on Chloride penetration into concrete, Saint Remy-les-Chevreuse, pp. 10-16, 15-18 October ,1995 5) 35 No.1 pp.1843-1848 2013.7 6) 34 No.1 pp.832-837 2012.7 7) NDIS 3433 2018 8) Vol.32, No.1, pp.785-790, 2010 9) 28 pp.129-132, 2016