第 54 卷 第 4 期
2019 年 8 月
JOURNAL OF SOUTHWEST JIAOTONG UNIVERSITY
Vol.54 No.4 Aug. 2019
ISSN - 0258-2724 DOI:10.35741/issn.0258-2724.54.4.22
Research article
Electrical and Electronic Engineering
C
OUMARIN
4
L
ASER DYE
/PVP
P
OLYMER
/A
L
2O3
N
ANOPARTICLE
T
HIN
F
ILMS
I
NDUCED BY
E
R
:
YAG
L
ASER
R
ADIATION
Mithaq M. Mehdy Al- Sultani
Department of Physics, College of Education for Girls, Kufa University, Iraq mithaq.alsultani@uokufa.edu.iq
Abstract
In this paper, thin films were made of 10-4 mol/l of Coumarin 4 laser dye dissolved in ethanol, 0.3 g of PVP polymer and 0.04 g of Al2O3 nanoparticles. The electrical properties such as dielectric constant, phase velocity of laser photons inside the film, the film permittivity and permeability, medium impedance, electrical conductivity, skin depth, the electrical response time, magnetic susceptibility and dielectric time have been studied for the prepared thin films at two different Er:YAG laser wavelengths of 1645nm and 1617 nm based on various laser powers in one time and with changed laser exposure periods in the other time. It is observed that the highest laser power consolidates each value of dielectric constant, dielectric time, electrical conductivity, the electrical response time, magnetic susceptibility and the permeability of film because of increasing the abundant energy in films within laser irradiation. The most important conclusion is that the electrical properties were much higher for the prepared thin films irradiated with 1645nm Er: YAG laser wavelength than for those irradiated with 1617nm laser wavelength.
Keywords: Coumarin4 dye, Er:YAG laser, Al2O3 nanoparticles, electrical properties.
摘要 : 物联网(IOT)是伊拉克新兴领域,已引起研究兴趣。其中一个最重要的方面涉及如何约束物联网以 加强伊拉克的医疗保健。该研究在计算机科学与信息技术学院进行。它讨论了不使用物联网管理伊拉克医 疗保健中危及生命的危急情况的影响。在这项研究中,我们使用通过巴士拉随机问卷调查收集的真实数据 ,考虑了物联网技术,特别是 DRONE 救护车。使用 SPSS 版本 0.23 分析数据。在研究结束时,我们确定了 可能影响物联网技术使用的因素:意识,安全性,成本,政府支持,协作和专业行为。结果表明,协作影 响了物联网技术的使用。 关键词: 香豆素 4 染料,激光,纳米粒子,电学性质。
I.
I
NTRODUCTIONCoumarin laser dye of C9H6O2 chemical formula stands for an aromatic biological
chemical compound in the benzopyrone category, although it can as well be classified as lactones subclass. It is an organic substance found in numerous plants and a colorless crystalline constituent in its typical state. It has physical
properties of 146.15 g·mol−1 molar mass, 0.935 g/cm3 density, 71°C melting point, 301.71°C boiling point and −82.5×10−6 cm3/mol magnetic susceptibility. It is feasibly employed as a successful laser intermediate for dye lasers [1-4].
Aluminum oxide nanoparticles (Al2O3) can be found as sphere-shaped or approximately spherical nanoparticles, or as undirected or oriented fibers. It has the properties of tiny particles/fibers ranging from 2 to 10 nm, great particular surface area (>100 m2/g), significant inadequacy of the material surface and specific structure of the nanoparticles in terms of the pore volume and size, phase composition, crystallinity degree, structure, and formation of the surface. It can be used in capturing hydrocarbon impurities from the air, in taking out fluorine from various media, in the gases drying, as a sorbent of radionuclides from wastewaters of nuclear power plants, in fabricating ceramics and composites including composite metals of immense toughness, in strengthening filler, fire resistance, and anti-friction features with insulating features. As well as in these applications, it can be employed as a catalyst and catalysts carrier [5-8]. The polyvinylpyrrolidone polymer (PVP) is a water-soluble polymer made from the N-vinylpyrrolidone monomer. It has the chemical formula of (C6H9NO) n. Its physical properties include density of 1.2 g/cm3, the molar mass ranging from 2,500 to 2,500,000 g·mol−1 and a melting point ranging from 150 to 180°C. It can be used in medical fields as in a plasma volume expander for trauma victims and as a lubricant in some eye drops while it may be used in technical areas as metal quenching for membrane production, and as a stabilizing mediator in each inorganic solar cell [9-12].
In [3], fluorescence, absorption along with phosphorescence features were studied for a succession of coumarin laser dye combinations with methanol and polyelectrolyte polymethacrylic acid (PMMA) based water media. The amplified spontaneous emitting and lasing features of Coumarin 540 dye in different ten solvents were testified and it was found that C 540 dye solutions controlled within a rectangular quartz cuvette provide laser emission with fit resolved spaced modes as pumped under a 476 nm beam [14]. In [15], dyes under microwave irradiation were created in the solid phase and solvent-free approaches with 80% yield that represents a huge and extraordinary percentage [15].
In this research, parametric investigations into the effect of Er:YAG laser power and laser exposure time in the electrical properties are
presented in terms of dielectric constant, phase velocity of laser photons inside the film, the film permittivity and permeability, medium impedance, electrical conductivity, skin depth, the electrical response time, magnetic susceptibility and dielectric time, for thin films of 10-4mol/l for Coumarin 4 laser dye dissolved in ethanol, 0.3 gm of PVP polymer and 0.04 gm of Al2O3 nanoparticles, under 1645 and 1617 nm wavelengths in the case of Er:YAG laser.
II. THEORY
The absorbance (A) describes the number of absorbed photons by molecules that can be evaluated based on the given transmittance (T) by [16-17]:
A = 1 − log 1
T (1).
Laser photons also suffer from refraction, therefore the refractive index (n) of material will be changed with incident laser intensity variation. Accordingly, T is calculated by [16-17]:
T = n2n2+1… … … . 2 − a , where 𝑛 can be calculated by [17-18]:
n = 2+ 4−4T2T 2… … … 2 − b . The material has several electrical properties which have been affected by the laser light. The dielectric constant (ε) refers to that portion of material which has no free charges based on the following equations [17-18]:
n = Km ε … … … . . 3 ,
where Km = 1 for rear end media so that dielectric constant makes [19-20]:
ε = n2… … … … … . 4 .
The laser has a specific velocity (vph) when
across within crystal [19-21] that can be calculated by:
vph = c
n… … … . . 5 ,
where C is speed of light in the vacuum [ 19-20]. Also, it can be computed by:
vph = 1
where Є stands for the material permittivity and can be defined as the ratio of the electric flux density to the electric field strength and μ is the material permeability which is known as the ratio of the magnetic flux density to the magnetic field strength. It can also be written as [19-22 ]:
ϵ = ε ϵ
o… … … 7 ,
where Єo is the free space permittivity that equals to 8.854x10-12 F/m and μ can also be calculated according to the following equation [19-21]:
μ = 1
ϵvph2 … … … 8 .
The electromagnetic field of laser suffers from resistivity (Z) within across media and it can be written as [22]:
Z = μ
ϵ… … … . 9 .
Electrical conductivity 𝜎𝐸 is a property of a
material that quantifies the straightforwardness of charge flow inside the material along an applied electric field. It can be calculated by [21]:
ςE = 1
Z .ℓ … … … 10 ,
where ℓ is the material thickness. An electromagnetic field of the laser reaches a specific depth in the material which intercepts its way. This depth is known as skin depth (δ) and it can be evaluated by [ 21 ]:
δ = μ ς2
E ω … … … … …. 11 , where ω is the incident wave frequency. Both 𝜎𝐸 and permittivity Є of any media were
continued for a specific time which known as electrical response time tE [21]:
tE = ϵ
ςE… … … 12 . Electrical susceptibility (χ) is a material quantity that measures the extent of polarization in the material per unit field [21]:
χ = ε−1
4 π … … … . 13 .
The induced electronic polarization (Pin) is the displacement of the electron cloud of an
atom relative to the positive nucleus. The Pin can
be given as [21]:
Pin = N q χ … … … … . . 14 ,
where N is the quantity of material molecules and q is the number of anti-bond electric charges emitted by each molecule multiplied by the charge. Molar polarization of a material (ρp) can be given by [21]: ρp = ε−1 ε+2 . MW d … … … . 15 ,
where 𝑀𝑊 is the material molecular weight,
and 𝑑 is the material density. Wave number (𝐾) stands for a magnitude of the wave vector in the material as shown in the following equation [21]:
K = ω
c ε … … … 16 .
In the beginning of laser or any electromagnetic wave applied to any material, nothing happened for its electrical properties for a period called as dielectric time τdi e [21]:
τdi e = ℓ εc … … … …. … 17 .
III. M
ETHODSA. Preparation of the samples
About 0.001 g of Coumarin 4 laser dye was dissolved in 10ml of Ethanol. And 0.3 g of PVP polymer was dissolved in the same dye solution using the magnetic stirrer to form a homogeneous solution. After that, 0.04 g of Al2O3 nanoparticles were added to the solution mentioned above.
The colloids of Coumarin 4 laser dye, PVP polymer, and Al2O3 nanoparticles were applied on the glass slide, to make a uniform thin film having thickness of 2.5mm.
B. Sample irradiation by laser
Er: YAG laser type based on IEC EN 60825-1: 2007 standard supplied from DNA Laser Technology Corporation, was used to irradiate these thin films by two different wavelengths of 1645 and 1617 nm; laser power at 0.2, 0.4, 0.75, 1.4, 1.75, 2.5 and 3 Watt of Er: YAG was applied for both wavelengths, to irradiate the prepared samples. The required times to irradiate the prepared samples by Er: YAG laser were also varied as 20, 40, 60, 80, 100,120 and 140 s.
The absorbance was measured for all prepared samples by varying the laser power and laser exposure time at 1645 and 1617 nm laser wavelengths using Spectrophotometer based on Scinco, Mega -2100 Korean version
D. XRD tests
To examine the material structure, a diffractometer was used as an evaluating device to study the structure of the prepared samples depending on the created scattering pattern as a beam of one of X-rays or neutrons cooperating with it.
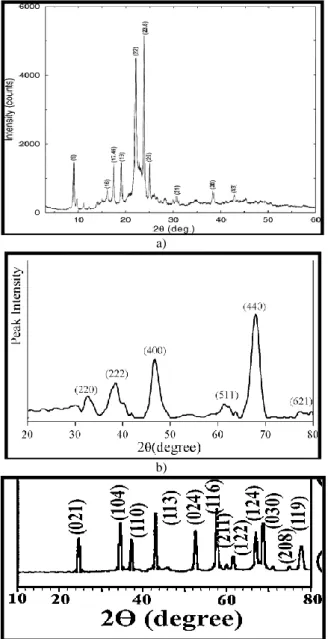
The X-Ray diffractometer used in this research is based on ADX-2700 X-ray powder diffraction model. Figure 1 illustrates the X-ray diffraction pattern for Coumarin 4 laser dye, Al2O3 nanoparticles and one of the prepared thin films.
a)
b)
c)
Figure 1. The X-ray diffraction pattern for (a) Coumarin 4 laser dye; (b) Al2O3 nanoparticles and (c) prepared thin film.
IV. R
ESULTSThe optical parameters such as absorbance A, transmittance T and refractive index n for 10 -4
mol/l of Coumarin 4 laser dye, 0.3 g of PVP polymer and 0.04 g of Al2O3 nanoparticle thin films induced by Er:YAG laser radiation at different values of laser power were calculated based on equations (1-2) depending on their optical spectrum. These results are listed in Table 1.
The electrical parameters such as 𝑡𝐸, 𝜎𝐸, 𝑡𝑑𝑖𝑒
and 𝜇 for 10-4 mol/l of Coumarin 4 laser dye, 0.3 g of PVP polymer and 0.04 g of Al2O3 nanoparticle thin films induced by Er:YAG laser radiation at different values of laser power were calculated using equations (12, 10, 17 and 8), respectively. The electrical results are detailed in Table 2.
Figure 2 illustrates the behavior for both medium impedance 𝑍 and dielectric constant𝜀 calculated according to equations 4 and 9 using different laser powers at two different Er: YAG laser wavelengths.
Figure 2. The medium impedance 𝑍 and dielectric constant using different laser powers at two different Er: YAG laser
wavelengths
Figure 3 depicts the performance of phase velocity of laser photons inside the film and the film permittivity calculated according to equations 5 and 7 using different laser powers at two different Er: YAG laser wavelengths.
Figure 3. The phase velocity of laser photons inside the film 𝑣𝑝ℎ and film permittivity 𝜖 using different laser powers
at two different Er: YAG laser wavelengths
Figure 4 describes the behavior of skin depth and magnetic susceptibility using different laser powers at two different Er: YAG laser wavelengths.
Figure 4. Skin depth 𝛿 and magnetic susceptibility χ using different laser powers at two different Er: YAG laser wavelengths
The optical parameters such as A, T and n for 10-4mol/l of Coumarin 4 laser dye, 0.3 g of PVP polymer and 0.04 g of Al2O3 nanoparticle thin films induced by 0.5 Watt of Er:YAG laser radiated power at different values of laser exposure time were determined depending on their optical spectrum. These results are listed in Table 3. The electrical parameters such as 𝑡𝐸, y
𝜎𝐸, 𝑡𝑑𝑖𝑒 and 𝜇 for 10-4mol/l of Coumarin 4 laser
dye, 0.3 g of PVP polymer and 0.04 g of Al2O3 nanoparticle thin films induced by 0.5 Watt of Er:YAG laser radiation at different values of laser exposure time are detailed in Table 4.
Table 1.
The optical parameters for 10-4mol/l of Coumarin 4 laser dye, 0.3 g of PVP polymer and 0.04 g of Al
2O3 nanoparticle thin films induced by Er:YAG laser radiation at different values of laser power
Laser power (W) Absorbance A Transmittance T Refractive index n 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 =𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 0.2 0.8 0.78 0.158 0.165 12.578 12.038 0.4 0.81 0.80 0.154 0.158 12.909 12.578 0.75 0.83 0.81 0.147 0.154 13.431 12.909 1.4 0.86 0.83 0.138 0.147 14.423 13.531 1.75 0.88 0.86 0.131 0.138 15.201 14.423 2.5 0.90 0.89 0.125 0.128 15.937 15.560 3 0.92 0.90 0.120 0.125 16.606 15.937 Table 2.
The electrical parameters for 10-4mol/l of Coumarin 4 laser dye/ 0.3 g of PVP polymer/0.04 g of Al2O3 nanoparticle thin films induced by Er: YAG laser radiation at different values of laser power
Laser power (W)
Electrical response time 𝒕𝑬 x10-12 (S) Electrical conductivity 𝝈𝑬 x10+3 (Ohm.m)-1 Dielectric time 𝒕𝒅𝒊𝒆 x10-11(S) Magnetic permeability 𝝁 x10-7 (H.m-1) 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 0.2 10.526 10.078 0.133 0.127 10.481 10.031 12.610 12.680 0.4 10.838 10.534 0.136 0.132 10.754 10.481 12.608 12.672 0.75 11.328 10.806 0.143 0.136 11.275 10.757 12.604 12.665 1.4 12.026 11.320 0.153 0.143 12.019 11.275 12.567 12.658 1.75 12.695 12.041 0.161 0.152 12.667 12.019 12.561 12.608 2.5 13.295 12.981 0.169 0.165 13.280 12.966 12.553 12.593 3 13.942 13.280 0.175 0.169 13.838 13.280 12.649 12.564 Table 3.
The optical parameters for ( 10-4mol/l of Coumarin 4 laser dye/ 0.3 gm of PVP polymer / 0.04 gm of Al2O3nanoparticles) thin films induced by 0.5 W of Er: YAG laser radiation at different values of laser exposure time
Laser eposure time (S) Absorbance A Transmittance T Refractive index n 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 20 0.82 0.81 0.151 0.154 13.169 12.909
40 0.83 0.82 0.147 0.151 13.531 13.169 60 0.85 0.84 0.141 0.144 14.113 13.816 80 0.87 0.85 0.134 0.141 14.858 14.113 100 0.89 0.87 0.128 0.134 15.560 14.858 120 0.91 0.90 0.123 0.125 16.198 15.560 140 0.94 0.93 0.114 0.117 17.486 17.035 Table 4.
The electrical parameters for 10-4mol/l of Coumarin 4 laser dye/ 0.3 g of PVP polymer/0.04 g of Al2O3nanoparticle thin films induced by 0.5 W of Er: YAG laser radiation at different values of laser exposure time
Laser exposure
time (S)
Electrical response time 𝒕𝑬 x10-12 (S) Electrical conductivity 𝝈𝑬 x10+3 (Ohm.m)-1 Dielectric time 𝒕𝒅𝒊𝒆 x10-11 (S) Magnetic permeability 𝝁 x10-7 (H.m-1) 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟒𝟓 𝒏𝒎 𝝀𝒍𝒂𝒔𝒆𝒓 = 𝟏𝟔𝟏𝟕 𝒏𝒎 20 11.012 10.806 0.139 0.136 10.974 10.757 12.650 12.665 40 11.312 11.012 0.143 0.139 11.275 10.974 12.638 12.650 60 11.793 11.544 0.149 0.146 11.760 11.513 12.627 12.631 80 12.415 11.793 0.157 0.149 12.381 11.760 12.620 12.627 100 12.997 12.407 0.164 0.157 12.966 12.381 12.609 12.613 120 13.509 13.280 0.171 0.169 13.498 13.280 12.588 12.564 140 14.582 14.195 0.185 0.180 14.571 14.195 12.580 12.552
Figure 5 illustrates the behavior for both 𝑍 and 𝜀 calculated according to equations 4 and 9 using 0.5 Watt of laser power at two different Er: YAG laser wavelengths for different exposure periods.
Figure 5. Medium impedance 𝑍 and dielectric constant using 0.5 Watt of laser power at two different Er: YAG laser
wavelengths for different exposure periods.
Figure 6 clarifies the performance for 𝑣𝑝ℎ and
𝜖 according to equations 5 and 7 using 0.5 Watt of laser power at two different Er: YAG laser wavelengths for different exposure periods.
Figure 6. Phase velocity of laser photons inside the film 𝑣𝑝ℎ and, the permittivity of film 𝜖 using 0.5 Watt of laser power
at two different Er: YAG laser wavelengths for different exposure periods.
Figure 7 describes the behavior for 𝛿 and 𝜒 calculated according to equations 11 and 13 using 0.5 Watt of laser power at two different Er: YAG laser wavelengths for different exposure periods
Figure 7. Skin depth 𝛿 and magnetic susceptibility χ using 0.5 Watt of laser power at two different Er: YAG laser
wavelengths for different exposure periods.
V. D
ISCUSSIONFigure 2 shows the effect of Er: YAG laser power on both medium impedance and dielectric constant based on 1645 and 1617 nm of two different Er: YAG laser wavelengths. The medium impedance was decreased with the increasing Er: YAG laser power. At the same time, the dielectric constant has been raised with laser power increasing. This behavior is determined by the highest laser powers releasing more electrons from atomic orbitals which induce
electrical conductivity and reduce medium impedance. The highest anti-bond electrons cause more reduction in the transmittance of laser medium and more increase in both absorbance and refractive index for that medium as shown in Table 1.
The increase in ϵ and the decrease in vph with
increasing Er: YAG laser power are illustrated in Figure 3. These changes are explained by the fact that the highest laser power makes the medium more sensitive to the applied electric field with the highest values of ϵ, 𝑡𝐸 and 𝜎𝐸as shown in
Table 2.
Figure 4 shows the increase in 𝜒 and decrease in 𝛿 with increasing Er: YAG laser power. This can be related to the high electricity level that was found in the media irradiated by laser photons, which induces the magnetic susceptibility and permeability, as shown in Table 2. The thermal diffusion through the surface layers of the medium under the laser is the main reason to reduce the skin depth with increasing applied laser power.
Figures 5, 6 and 7 explain the effect of laser exposure time on the electrical and magnetic properties for laser medium as in the thin film of Coumarin 4 laser dye. In accordance with passing time of applied laser, electrical parameters such
as 𝜀, 𝜎𝐸 ,𝑡𝐸 , 𝑡𝑑𝑖𝑒 and the magnetic parameters
such as 𝜒 and t 𝜇 increased as shown in Table 4, and all values of 𝑍 , vph, 𝛿 and 𝑇 properties decreased. The increase in electrical and magnetic properties is attributed to the highest absorptivity, which consolidates the electrical conductivity by additional released electrons making the medium more refractive as indicated by𝑛 and absorbance 𝐴 values in Table 3. A reduction in the values of 𝑍, 𝑣𝑝ℎ, δ and 𝑇 with
increased exposure time is noticed since the thermal waves are generated in the surface layers of laser dye medium. It was evident from Figures 2-7 and Tables 1-4 that all the optical, electric and magnetic properties for the thin film at 1645 nm are more significant than those at1617 nm. This can be attributed to the fact that the larger laser wavelength induces the molecules in the nearest states of levels separated by small differences and excites them by absorption process. Consequently, all electrical properties related by optical properties are consolidated.
VI. C
ONCLUSION AND FUTURE WORKSIt can be concluded that the laser dye medium is more sensitive for applied laser power and laser exposure time, where the electrical and magnetic properties are in high quality after laser
radiation. The generation of thermal waves through the surface layers of the laser medium impedes the laser spread in the deepest layers and the excitation of molecules in them. In a nut shell, Er: YAG laser wavelength of 1645 nm is more suitable and significant for studying the optical, electrical, and magnetic properties for laser dye media as compared to the type of 1617 nm wavelength. The performance of above lasers can be analyzed further as future work by means of nanotechnology techniques.