Effects of an Indonesian Medicinal Plant, Curcuma xanthorvhiza
Roxb., on the Levels of Serum Glucose and Triglyceride,
Fatty Acid Desaturation,
and Bile Acid Excretion in
Streptozotocin-induced
Diabetic Rats
Sedarnawati
Yasni,
Katsumi Imaizumi
and Michihiro
Sugano
Laboratory of Nutrition
Chemistry,
Department of Food Science and Technology,
School of Agriculture
(46-09),
Kyushu University,
Fukuoka 812, Japan
Received June 10, 1991
To study the beneficial effects of traditional Indonesian foods on sugar and lipid metabolism, streptozotocin-induced diabetic rats were fed on purified diets containing 5%of either cellulose as a control or four kinds of Indonesian plants. One of them, Curcuma xanthorrhiza Roxb., improved the
diabetic symptoms such as growth retardation, hyperphagia, polydipsia, elevation of glucose and triglyceride in the serum, and reduction of the ratio of arachidonate to linoleate in the liver phospholipids. C. xanthorrhiza specifically modified the amount and composition of fecal bile acids. Significance of these findings was discussed in the light of the improvement of several diabetic symptoms.
In Indonesia there are manyvegetables and
herbs that have been widely used as traditional
medicinal
plants
since ancient times. Dharma
described
145 kinds of traditional
medicinal
plants based on their pharmacological,
clinical,
nutritive,
and economic values.1} Some of them
were commonlyused to prevent and treat
several
chronic
diseases,
such as diabetes
mellitus and heart ailments with less scientific
diagnoses.2>3)
Amongthese medicinal plants in Indonesia,
rhizomes of Curcuma xanthorrhiza Roxb.
{Temulawak in Indonesia) and Zingiber
aroma-ticum Val. (Lempuyang in Indonesia) are used
as vegetables and beverages.4'5) The seeds of
Parkia speciosa Hassk. {Petal in Indonesia) and
Pithecellobium jiringa Prain. (Jengkol in
Indonesia) are Indonesian popular food, eaten
as side dishes
or served as vegetables.4'5)
To
examine the active principles
of these medicinal
plants,
it is
important
to investigate
their
physiological
functions
when they are eaten as
food ingredients.
In this study we have examined the effects
of these medicinal plants on feeding habits,
growth, and metabolic parameters of
strepto-zotocin-induced
diabetic
rats.
This
study
demonstrates
the
beneficial
effects
of
C.
xanthorrhiza
on several diabetic symptoms.
Materials
and Methods
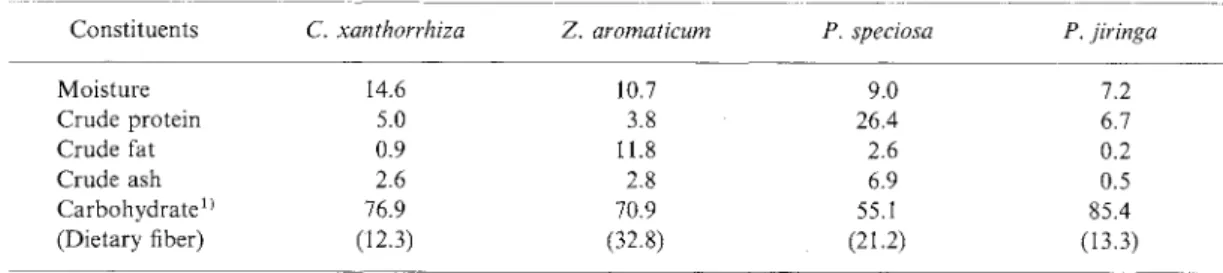
Preparation of Indonesian medicinal plants. Four kinds of Indonesian plants, C. xanthorrhiza, Z. aromaticum, P.
speciosa and P. jiringa were purchased in a traditional market in West Java, Indonesia. Seeds of P. jiringa and P. speciosa, and rhizomes of C. xanthorrhiza and Z. aromaticum were freeze-dried, milled, and sifted through a 100-mesh sieve. The powdered samples were analyzed
for their chemical composition6) including dietary fibers7'
(Table I).
Experimental animal. Male Sprague-Dawley rats, 6
weeks old and obtained from Seiwa Experimental Animal Co. (Fukuoka), were housed individually in stainless steel
cages with wire-meshed bottoms in an air-conditioned
room (23 + 1°C) with a controlled dark-light cycle (lights on 08 :00-18 :00). The rats were freely given water and commercial pellets (Type NMF, Oriental Yeast Co.,
Tokyo) for 2 weeks, and were then starved for 16 hr before
injection
of streptozotocin
(60mg/kg body weight, Sigma
Chemical Co., MO) into the femoral vein under light diethyl ether anesthesia.8) The animals were then fed forTable II. Composition of Basal Diet Ingredients Amount
(g/lOOg diet)
Casein" gafflower o\\b a-Corn starchc Mineral mixture** Vitamin mixture6Choline Bitartratee
DL-Methionine-^ Supplements0 Sucroseb 30 5 15 3.5 1.0 0.2 0.3 5 50Vitamin-free (Wako Pure Chemicals Ltd., Tokyo). Obtained from local market.
Nippon Shokuhin Kakoh Ltd., Aichi.
AIN-76 mixtures, purchased from Oriental Yeast
e
/
g
Co., Tokyo).
Katayama Chemical Ltd., Osaka.
Nacalai Tesque, Inc., Kyoto.
Either cellulose (Type E, Toyo Roshi Kaisha, Ltd., Tokyo), or 4 kinds of Indonesian plants.
2 weeks on an experimental diet containing 5% of either cellulose as a control or Indonesian plants, which were added to the basal diet as shown in Table II. The body weight and feed and water intake were recorded daily. The rats were killed by decapitation at 10 :00, and the blood serum was prepared by centrifugation. The liver and cecum were excised immediately, frozen with liquid nitrogen, and kept at -40°C until analyzed. Feces were collected for 2 days before killing.
Analytical procedure. Serum and liver lipids were
extracted in a chloroform-methanol mixture (2 : 1, v/v),9)
and analyzed for cholesterol, triglyceride, and phospho-lipid as described previously.10) The total phospholipid fractions in the liver were separated by thin-layer chromatography, and the fatty acid composition was analyzed by gas-liquid chromatography.1 X) Serum glucose was measured with a commercially available kit (Glucose Test, Wako Pure Chemical Co., Osaka). Fecal acidic and
neutral steroids were measured by gas-liquid
chromato-graphy on 3% AN-600 and 3% OV-17 columns,
respectively, as described previously.12) Short-chain fatty acids in the cecumwere measuredas reported elsewhere. 13)
Statistics. Data were analyzed by Duncan's multiple
range test preceded by analysis of variance (ANOVA),and by Student's t test.
Results
Addition
of
Indonesian
plants
exerted
different
affects
on food intake
and water
consumption in rats treated with streptozotocin
(Fig. 1). These values were markedly higher
than those non-diabetic rats with comparable
body weight, less than 20g offood intake and
20g of water consumption.
The food intake
and water consumption, however, were
signif-icantly lower in rats fed C. xantkorrhiza
than
in those
fed cellulose
or other
Indonesian
plants.
There was no significant
difference
in
the final body weight among the groups. The
relative liver weight did not differ among the
groups (data not shown).
As shown in Fig. 2, C. xanthorrhiza
sup-pressed elevation of serum glucose and
triglyc-eride significantly as compared to cellulose and other Indonesian plants. P. jiringa also
decreased the serum triglyceride level as
com-pared to cellulose.
The level of serum
choles-terol and phospholipid
was not influenced by
the supplements.
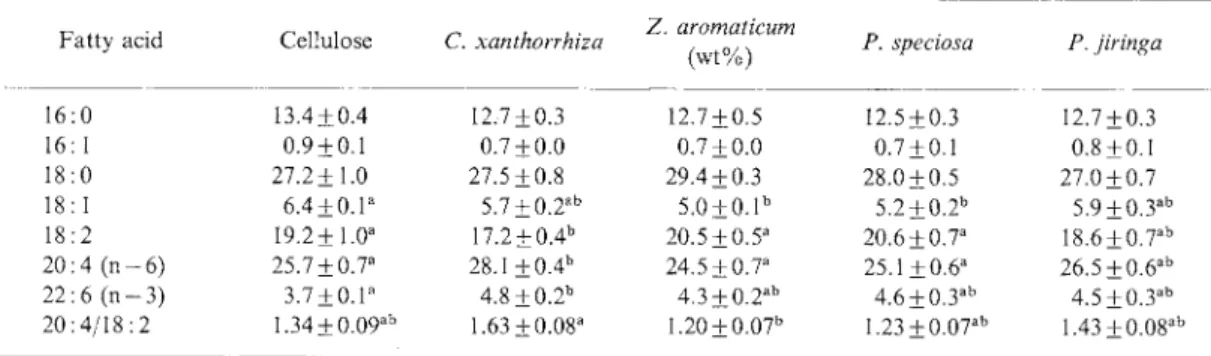
AmongIndonesian plants, C. xanthorrhiza
slightly but significantly
increased the
propor-tion of arachidonic acid (20 : 4), and decreased
linoleic acid in the liver phospholipids, as compared to cellulose (Table III). Thus, theFig. 1. Body Weight, Food Intake, and Water Consumption in Rats Fed Indonesian Plants.
The bars show mean+S.E. for 5 rats per group. abDifferent superscript letters denote significant difference
at/?<0.05.
Fig. 2. Serum Lipids and Glucose in Rats Fed
In-donesian Plants.
The bars show mean+S.E. for 5 rats per group. ^Dif-ferent superscript letters denote significant difference
at /?<0.05.
desaturation index (ratio of 20:4 to 18:2)
tended to be higher in rats fed C. xanthorrhiza
than in those fed cellulose or other supple-ments.
Fecal weight tended to be lower in all groups
of rats fed Indonesian plants, as compared to
celluose (Table IV). Excretion of total fecal
neutral sterols was lower in rats fed P. jiringa
than in those fed other diets. The excretion oi
coprostanol
was lower in rats fed Indonesian
plants except for P. speciosa, as comparedto
cellulose. In comparison with cellulose group,
excretion of cholesterol was greater in the C
xanthorrhiza group, and lower in rats in the P.
jiringa group. Thus, the ratio of coprostanol
to cholesterol
was markedly low in rats fed C.
xan thorrhiza.
As shown in Table V, C. xanthorrhiza,
as
compared to cellulose,
significantly
decreased
fecal total bile acids. The composition of bile
acids were specifically
modified by Indonesian
plants,
in particular
C. xanthorrhiza,
in which
the ratio of primary to secondary bile and
(deoxycholic acid/cholic acid and lithocholic
acid/chenodeoxycholic
acid) was exceptionally
low. P. speciosa and P. jiringa also modified
the ratio of primary to secondary bile acids.
As shown in Fig. 3, C. xanthorrhiza, as
compared to cellulose,
tended
to increase
the
mass of cecumacetate, and decrease that of
propionate
and ^-butyrate.
Cecum contents
increased significantly
in rats fed C.
xanthor-rhiza (data not shown).
Discussion
This
study
was carried
out to examine
whether
Indonesian
plants,
which
are
com-monly eaten and often
used as traditional
medicines, have a beneficial
effect when given
to diabetic rats. Diabetic animals have been
reported to show the following behavioral and
metabolic disorders:
Values are mean+ S.E.M. for 5 rats pergroup. ab Different superscript letters denote significant difference at/? <0.05. Table IV. Fecal Weight and Neutral Steroids Excretion in Rats Fed Indonesian Plants
Dietary groups Feces weight Coprostanol Cholesterol Total
(g/day)(mg/day) Coprostanol/ Cholesterol Cellulose
C. xanthorrhiza
Z. aromaticum
P. speciosa
P. jiringa
3.08±0.13a 2.29+0.343 2.31±0.16b 0.18+0.1lb 2.63±0.18ab 0.95±0.24c 1.54+0.17c 2.52±0.68a 1.13+0.10c 1.05+0.25c 3.04+0.47a 5.32±0.77ab 6.42±0.96b 6.50± 1.00b 2.64+0.213 3.59±0.20a 5.17± 1.30ab 7.69± 1.46b 1.17+0.11° 2.22+0.33c 0.75+0.093 0.03±0.10b 0.36±0.12c 0.49±0.13ac 0.90+0.19acValues are mean + S.E.M. for 5 rats per group. ab Different superscript letters denote significant difference at/> < 0.05. Table V. Fecal Excretion of Bile Acids in Rats Fed Indonesian Plants
D -1 -a i) r-ll 1 ^ / /à" Z.aromaticum . ...
Bile acids ' Cellulose C. xantnorrniza , , -, -, P- speciosa P. jiringa
(mg/day) y J 6 Deoxy Cholic Litho Cheno Hyo+Urso a- +a>Muri ^-Muri Total Deoxy/Cholic Litho/Cheno 2.11±0.24a 0.18+0.093 0.50+0.06a 0.42±0.05ab 0.50+0.10c 0.51 +0.063 0.15+0.023 4.38+0.433 ll.7±4.3a 1.19+0.08a 0.24±0.03b 1.23±0.22b 0.18+0.05b 0.36±0.07ab 0.04±0.02b 0.50+0.073 G.41±0.14ab 3.10±0.14b 0.20±0.04b 0.50+0.10b 2.07+3.043 0.11±0.03a 0.19±0.05b 0.07±0.02a 0.23±0.08c 0.18±0.02b 0.37±0.10ab 3.21 ±0.43ab 18.8±3.2bc 2.71 ±0.89c 4.07± 1.20c 0.24±0.09a 0.57±0.09a 0.25±0.07b 0.39+0.103 0.72±0.24a 0.23 ±0.09ab 6.36+ 1.52ab 17.0+6.4c 2.28+0.35c 1.81±0.17ab 0.24±0.07a 0.43±0.05a 0.29±0.09b 0.32±0.06ac 0.53+0.093 0.45±0.llb 4.07±0.38a 7.54±0.8b 1.48+0.37c
Values are mean + S.E.M. for 5 rats per group. abc Different superscript letters denote significant difference at/? < 0.05.
1} Deoxy, deoxycholic acid; Cholic, cholic acid; Litho, lithocholic acid; Cheno, chenodeoxycholic acid; Hyo,
hyocholic acid; Urso, ursodeoxycholic acid; Muri, muricholic acid.
(1) growth retardation
in spite of
hyper-phagia, 14)
(2) polydipsia due to increased water loss
into urine,14)
(3)
elevation
of
serum glucose
due to
increased
gluconeogenesis
in the liver and/or
insufficient
peripheral use, 1 5)
(4) enhanced level of serum triglyceride
due
to increased production of chylomicron in the
intestine and/or insufficient action of peripheral
lipoprotein lipase, 16)Fig. 3. Cecum Short-Chain FAtty Acids in Rats Fee
Curcuma xanthorrhiza Roxb.
The bars showmean+S.E. for 5 rats per group.
activity
in several
tissues
due to insufficient
insulin
action,17'18)
and
(6) an expanded pool size of cholic acid
due to increased synthesis.19)
Among Indonesian plants tested in this
study, C. xanthorrhiza ameliorated
hyper-phagia, polydipsia, glucosulemia,
triglycer-idemia, and z!6-desaturation.
Growth
retarda-tion also tended to be improved by this plant.
C. xanthorrhiza appeared to decrease the pool
size of cholic of total bile acids, the mass of
cholic acid plus deoxycholic acid was lower in
rats given C. xanthorrhiza, as compared to
con-trol rats (2.29+0.27 vs. 1.47+0.24mg/day,
p< 0.05). These ameliorating effects on
diabetic symptoms were not observed for
other Indonesian plants, except for P. jiringa,
which was partly
effective
to decrease
the
serum triglyceride
level.
C. xanthorrhiza have been reported to
contain several active principles, such as
germacrone,20) curcuminoid,21'22) xanthorri-zol,23) and essential oils.22) Pharmacological
studies have shown that those principles in C.
xanthorriza exert effects antibacterial,24) anti-oxidant,25) antiinflammatory,20) antihepatox-ic,26) anti-rheumatic,27) and hypothermic ef-fects.23) None of the preceeding reports,however, have dealt with the beneficial
effects
of the principle(s)
in C. xanthorrhiza
for
dia-betic symptoms. Someabsorbable principle (s)
of C. xanthorrhiza,
may potentiate
the
re-sidual
insulin
action
even after pancreatic
/?-cells
were severely
injured
by
streptozotocin-treatment.14)
Therefore, it is warrant to stud>
whether C. xanthorrhiza increases the potential
of the insulin reaction with its receptor and/or
glucose
transporter.
14)
This
study showed that
Indonesian
plants
modified
the fecal
steroid
composition,
pre-sumably
due to the change
in the
activity
and/or population
of colonic
micro flora.28)
C
xanthorrhiza specifically suppressed the trans-formation of cholesterol to coprostanol, andprimary bile acid to secondary
bile acids.
These
effects
appeared
to be attributed
to some
antifungal
or bacteriocidal
principle(s)
in C.
xanthorrhiza.2^
This
principle(s),
however,
appears to be less effective as an antibiotic
and
may not have indiscriminately
modified the
micro flora, since the production of cecum
acetic acid, which is mainly produced by
non-putrevactive bacteria,29) tended to be
increased
by feeding
C. xanthorrhiza.
However,
the C. xanthorrhiza
seems to influence the
population of colonic microorganisms, since
the number of microorganisms capable of
synthesizing
the specific
7a-hydroxylase,
which
is responsible for microbial transformation of
cholic acid and chenodeoxycholic acid to their
corresponding secondary bile acids, is onlyabout 104-106 organism per gram weight
feces.30)
Although
it is
totally
unsolved
whether
colonic micro flora mayhave beneficial effects
on diabetic
hosts, a change of the microbial
ecosystem in the digestive tract induced by a
low concentration of antibiotics has been
reported to be beneficial for increasing feed
efficiency
for broiler
chickens.31)
Alteration
of
colonic environment induced by feeding C.
xanthorrhiza, therefore, maybe relevant to
relatively
better body weight gain in rats fed
C. xanthorrhiza, as compared to cellulose and
other Indonesia plants.
In summary, this
short-term
experiment
showed that rhizomes of C. xanthrrhiza
added
to diets
exert
beneficial
effects
on several
metabolic disorders in streptozotocin-induced
diabetic rats. Anextension of these findings to
non-insulin dependent diabetic models, which
is a more commontype of diabetes, deserves
attention.2) O. T. Tampubolon, "Medicinal Plants (in In-donesian)," by Bharata Karya Akasara Press, Jakarta, 1981, pp. 78-79 and pp. 117-119.
3) Y. S. Kasahara and S. Mangunkawatja, "Medicinal Herb Index in Indonesia," by P. T. Eisai Indonesia, Jakarta, 1986, pp. 153-154 and pp. 343-348.
(1971).
19) M. T. R. Subbiah and R. L. Yunber, Biochem. Biophys. Res. Com., 124, 896 (1984).
20) Y. Ozaki, Chem. Pharm. Bull, 38, 1045 (1990).
21) I. Lubis, Annales Bogorienses, IV, 219 (1968). 22) O. B. Liang, Y. Apsarton, T. Widjaya and S. Puspa, M. L. Perry, "Medicinal Plants of East and South
East Asia, Attributed Properties and Uses," MIT Press, Cambridge United States and London, 1980,
pp. 221-222 and pp. 44(M44.
S. Setijati, L. Siti Harti Aminah, D. Eddy, S. Hadi
and L. Ischak, "Vegetables," by Bina Karya Press, 23) Bogor, 1981, pp. 77-78 and p. 83.
"Isolation, Identification Aspects of Curcuma
xan-thorrhiza Roxb. and Curcuma domestica Val.
Components (in Indonesian)," Proceeding of The
National Symposium on Temulawak, September 17, 1985, Bandung, Indonesia, 1985, p. 85.
M. Yamazaki, T. Maebayashi, N. Iwase and T. Kaneko, Chem. Pharm. Bull, 36, 2070 (1988).
6) Association ofOfficialAnalytical Chemists, "Official 24) T. Kasuge, H. Ishida and H. Yamazaki, Chem.
Methods of Analysis," 14th ed., ed. by M. Sidney, Pharm. Bull, 33, 1499 (1985).
TheAssociation ofOfficialAnalytical Chemists, Inc., 25) S. Toda, T. Miyase, H. Arichi, H. Tanizawa and Y.
Arlington, 1984, pp. 152-160.
N. G. Asp, C. G. Johansson, H. Hallmer and M. Siljestrom,V. Agric. Food Chem., 31, 476 (1983) A. Junod, A. E. Lambert, W. Stauffacher and A. E. Renold, /. Clin. Invest., 48, 2129 (1969).
J. Folch, M. Lees and G. H. Sloane-Stanley, /. Biol. Chem., 226, 497 (1957).
K. Imaizumi, Y-F. Lu and M. Sugano, Biochim.
Biophys. Ada., 917, 269 (1987).
K. Imaizumi, M. Sakono, K. Mawatari, M. Murata, and M. Sugano, Biochim. Biophys. Ada, 1005, 253
(1989).
M. Sugano, Y. Yamada, K. Yoshida, Y. Hashimoto,
Takino, Chem. Pharm. Bull, 33, 1725 (1985).
Y. Kiso, Y. Suzuki, N. Watanabe, Y. Oshima and
H. Hikino, Planta Med., 49, 185 (1983).
S. O. Deodhar, R. Sethi and R. C. Srimal, Indian J.
Med. Res., 71, 632 (1980).
I. A. Macdonald, F. D. Bokkenheuser, J. Winter, A. M. McLernon and E. H. Mosbach, /. LipidRes., 24,
675 (1983).
K. Itoh, N. Shirakami and T. Mitsuoka, in "Intestinal Flora and Bio-Homeostatis," ed. by T. Mitsuoka, Japan Scientific Society Press, Tokyo, 1989, p. 5. E. J. Stellwag and P. B. Hylemon, /. LipidRes., 20,
325 (1979).
T. Matsuo and M. Kimoto, Atherosclerosis, 72, 115 31) A. Kaukas, M. Hinton and A. H. Linton, /. Appl.
(1988). BacterioL, 64, 57 (1988).