DEVELOPMENT O F RUST FUNGI I N OAT LEAVES TREATED W I T H BLASTICIDIN S, A PROTEIN SYNTHESIS INHIBITOR Toshikazu TANI, Hiroyuki YAMAMOTO, Genichi KADOTA and Nakato NAITO
Introduction
Previous studies were carried out to elucidate changes in protein metabolism of oat leaves fol- lowing infection with the crown rust fungus. Experiments on in uztro protein synthesis supported by the E coli cell-free system and ribonucleic acid preparations from oat leaves as a template revealed that no obvious changes in the template activity of the preparation occurred during hypersensitive reaction, while the activity increased during susceptible infection(l7). This simultaneously suggested that the resistant reaction was not accompanied with pronounced changes in protein synthesis at quantitative level I n subsequent experiments with resistance responding leaves, howevcr, an increase of a few proteins probably of host origin was demonstrated by irnmunoelectrophoresis and disc electrophoresis of l*C-labeled proteins(20). I t was thus suggested that infection with an incompatible race did induce the de nouo synthesis of certain pro-
teins without remarkable quantitative alterations in the protein synthesis.
The aims of the forthcoming works are to confirm the occurrence of induced protein synthesis in resistant infection and to explore whether the induced synthesis is causally linked with the expression of host resistance For this purpose, we first planned to screen any specific protein synthesis inhibitor which blocks the resistance expression without any influence on the susceptible infection, and found that blasticidin S (BcS)(l*) fulfilled our purpose. This paper deals with the effect of BcS on the development of incompatible and compatible races of the crown rust fungus in oat leaves. The work was extended to behaviors of non-pathogenic rust fungi in BcS-, treated oat leaves
Part of the study was presented a t the symposium on current topics in plant pathology held a t Budapest, Hungary, in June 1975
Materials and Methods
Plant and fungus : Four cultivar s of oat (Auena ratzua L ) and two races of the crown rust fungus
(Puccznza coronata CDA. fa sp. auenae FRASER & LED ) were used Cultivars Shokan 1 and Bond were resistant to race 226 responding with hypersensitive necrosis (Infection type 0;) and suscep- tible to race 203 (Infection type 4) Hyuga-Kairyo-Kuro (HKK) was also resistant to race 226, but reacted with no hypersensitive necrosis (Infection type 0-l), and susceptible to race 203 (Infection type 4). Victoria 226-S(19) was susceptible to both races (Infection type 4)
Phragmzdzum rosae-multzJorae DIETEL, Puccinia agrofiri ELL et EV
,
P. allzi (DC.) RUDOLPHI, P. oenanther-rtolonzfeerae S 110, P. recondzta ROBERGE et DESMAZI~RES, Uromycer alofiecuri SEYM,, andU durur DIETEL were used as non-pathogens to oats These fungi produced no symptom on oat leaves (Infection type I).
Methods for growing oat seedlings and inoculating uredospores to primary leaves of 7-day-old seedlings were identical to those described previously(9).
96 Tech Bull Fac Agr Kagawa Univ specified, the stem cut-ends dctached with primary leaves were placed into 5
x
10-6 M aqueous solution of BcS for 4 hr prior to inoculation, and subsequently transferred into deionized water until harvesting samples BcS was most effective a t this concentration to reverse resistant to susceptibleMicroscopic observation: Development of the fungi was estimated under light microscope after staining the leaf samples with lactophenol cotton blue by the whole leaf clearing method of SHIPTON and BROWN(^^) Details have been described elsewhere(g.16)
Assay of antifungal activity: Leaf epidermis was removed from the inoculated sides of leaves, and 1 0 g of the stripped leaf segments were dipped into 20 ml of deionized water for 5 min to obtain the leaf exudate For partial purification of antifungal principal, the leaf exudate was condensed to dryness in vacuo and the residue was dissolved in methanol T o the methanol solu- tion eight volumes of ethylether were added and the resulting precipitate was collected as the crude preparation. The leaf exudate and crude preparation from 1.0 g leaves were made u p by de- ionized water to final volumes of 40 ml and 20 ml, respectively. Uredospores of race 226 were floated on these solutions and germinated for 16 hr a t 20°C to estimate the antifungal activity.
Results
1. Vegetative development of fungi in BcS-treated leaves
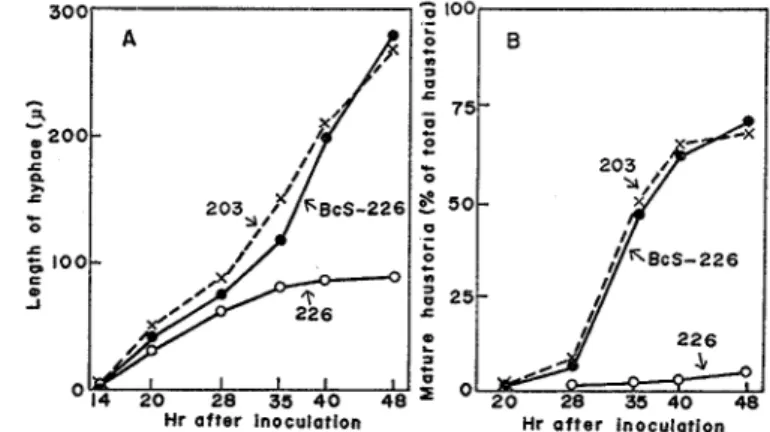
Fig. 1 shows the time courses of the elongation of infection hyphae and development of mature haustoria of normally incompatible race 226 in Shokan 1 leaves which were treated with BcS, and those of race 226 and compatible racc 203 in non-treated leaves I n non-treated leaves race 226 ceased the growth of infection hyphae between 35 and 40 hr after inoculation and produced essentially no mature haustoria I n leaves treated with BcS, however, hyphal growth and mature haustoria formation of race 226 were identical to those of race 203, indicating that the development of race 226 was completely altered to compatible type by BcS treatment.
Hr after lnoculatlon Hr a f t e r inoculation
Fig 1. Vegetative dekelopment of P coronata avenue in Shokan 1 oat leaves treated with BcS
A: Growth of infection hyphae B: Development of haustoria 22G=Incompatible race 226; 203= Compatible race 203; BcS-226=Kace 226 in BcS-treated leaves
BcS (5 x 10-6M) was supplied for 4 hr prior to inoculation
The same effect of'BcS was shown in the development of' race 226 in another resistant cultivar Bond, in which the hyphal length a t 48 hr after inoculation was 2.8 times longer than that in non-treated control leaves (Table I).. O n the other hand, no obvious effect of'BcS was detected
Vol. 27, No. 59 (1976) 97 on the hyphal growth of the compatible races including race 226 in
HKK
and Victoria 2 2 6 4 and race 203 in Shokan 1HKK
is resistant to race 226, but this cultivar does not restrict the development of vegetative growth of race 226, and therefore is actually susceptible until 72 hr after inoculation, when the initial stage of uredial differentiation occurs(9)Table 1. Growth of infection hyphae of P. coronata avenue in leaves of resistant and susceptible oat cultivars treated with BcS
Length of hyphae (p) in leaves
Inoculum Infection treated with
Cultivar race type Water BcS
Sholcan 1 226 0; 84 275
Bond 226 0; 77 215
HKK 226 0- 1 220 269
Victoria 226-S 226 4 24 1 266
Shokan 1 203 4 262 237
BcS (5 x M) was supplied for 4 hr prior to inoculation. Infection type was recorded by the disease appearance at 7 days after inoculation, and hyphal length was measured at 48 hr.
These results suggest that BcS supresses production of the factor which limits vegetative develop- ment of the fungus in resistant cultivars but does not affect the development in susceptible cul- tivars.
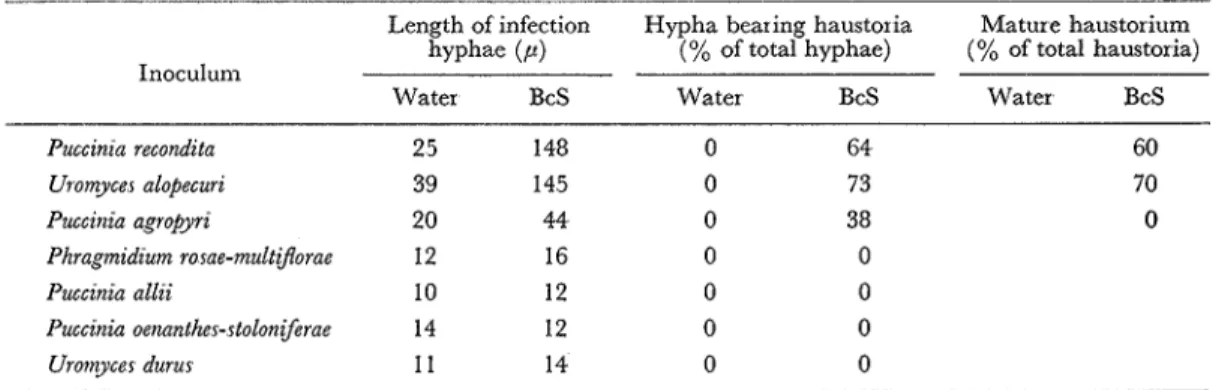
The development of non-pathogenic rust fungi in BcS-treated Shokan 1 was compared with that in non-treated control (Table 2). These fungi ceased the growth of infection hyphae within 20 hr after inoculation and produced no haustoria in the non-treated leaves I n BcS-treated leaves, however, two fungi P.. recondzta and U alopecuri grew well and produced haustoria abun- dantly, and 60 to 70% of haustoria were mature Production of immature haustoria was also observed for P. agropyrz. Neither stimulation of hyphal growth nor formation of haustoria was observed for the other five non-pathogenic rust fungi examined.
Table 2.. Development of non-pathogenic rust fungi in Shokan 1 oat leaves treated with BcS
-- -
Length of infection Hypha bearing haustoria Mature haustorium hyphae ( P ) (% of total hyphae) (% of total haustoria) Inoculum
Water BcS Water BcS Water BcS
Puccznza recondzta 25 148 0 64 Uromycer alopecurz 39 145 0 73 Puccznza agropyrz 20 44 0 38 Phragmzdzum rosae-multzjlorae 12 16 0 0 Puccznza allzi 10 12 0 0 Puccznia oenanther-rtoloniferae 14 12 0 0 Uromycer durur 11 14 0 0
BcS (5 x M) was supplied for 4 hr prior to inoculation, and fungal development was recorded 48 hr after inoculation
2. Uredial differentiation in BcS-treated leaves
98 Tech Bull. Fac. Agr. Kagawa Univ layers, and sporulation(g), was observed for race 226 in leaves treated with BcS. Race 226 normally proceeded no reproductive differentiation in Shokan 1 leaves However, in the leaves treated with BcS, 65% of mycelial colonies produced massing of hyphae on day 4 ; pseudoparen- chymatous layers were abundant on day 5 and some of these had already born immature uredo- spores; on day 7, uredospores were formed on most uredia (Fig 2-A) This time course of uredial differentiation was fairly identical to that of compatible race 203 in Shokan 1 (Fig 2-B). BcS treatment did not affect the uredial differentiation of compatible race 203 in Shokan 1 (Fig 2-C) Race 226 seldom produced uredia in the non-treated HKK leaves, but produced consider- able number of uredia in BcS-treated leaves (Fig 2-D)
-
U)3 4 5 6 7 3 4 5 6 7 3 4 5 6 7 3 4 5 6 7 D a y s after inoculation
Fig 2 Uredial diffelentiation of P coronata avenue in oat leaves treated with BcS A: Incompatible race 226 in BcS-treated leaves of Shokan 1
B: Compatible race 203 in water-treatcd control leaves of Shokan 1 C: Compatible race 203 in BcS-treated leaves of Shokan 1
D: Incompatible race 226 in BcS-treated leavcs of H K K
M=Massing of hyphae; P=Pseudoparenchymatous layers; S=Uredia bearing uredospores BcS (5 x 10-6M) was supplied for 4 hr prior to inoculation.
Thus, BcS enhanced the uredial differentiation of the normally incompatible race in the both cultivars BcS treatment, howevcr, failed in continuous stimulation of mature uredospore production in further incubation, probabIy because of its toxic effect on oat lcaves
3. Possible mechanism of BcS in blocking the resistance
Following experiments were carried out in order to assess whether the effect of BcS on the de- velopment of incompatible race 226 as described above was due to direct action of BcS on the fungus or to the blockage of host resistance by BcS
Experiment 1: Uredospores were floated on BcS solutions a t various concentrations and in- cubated at 25OC for 16 hr As shown in Fig 3, apparent inhibition of germination and germ tube elongation occurred a t 5
x
and 10-6 M, respectively, but no acceleration of the growth was observed at any concentration tested (10-5-10-7 M), despite that in Shokan 1 leaves the growth of infection hyphae and production of mature haustoria were stimulated a t 10-5-5x 10-7 M.
Experiment 2: Uredospores of race 226 were brushed on leaf surface of Shokan 1, then the
leaf epidermis was removed from mesophyll tissues and floated on 5 x 10-6 and 5 x 10-7 M BcS solutions or water At 48 hr later, the length of infection hyphae in BcS solutions was in the range of 19 4 to 24.6 p and similar to that in water. This suggests that growth of infection
Vol.. 27, No.. 59 (1976)
Concentration of BcS(M x 107)
Fig 3 U~edospore germination and vcgetativc dcvclopment of P coronata avcnae racc 226 on BcS solutions and in BcS-treated Shokan 1 oat leavcs, respectively
G = Germination; GT=Gcrm tubes; III=Infection hyphac; MH=Mature haustoria Urcdosporcs were floated on BcS solutions and gcrininatcd for 16 hr BcS was supplied to leaves for 4 hr prior to inoculation and thc lungal dcvclopmc~~t was rc~orded 48 hr aftcr inoculation
hyphac is not stimulatcd by BcS Since the hyphal length on the strippcd epidermis which was hanged in moist air was 162 p, it was apparent that the limitation of hyphal growth in water was not due to the lack of endogenous nutrients
The results in Experiments 1 and 2 may deny thc possibility that BcS directly stimulates the fungal growth
Experiment 3: Urcdosporcs of race 226 were gcrminated on 5
x
10-6 M BcS solution for 2 hr, then placed on Shokan 1 leaves I n another experiment, 5x
10-6 M BcS solution was sprayed on leaf surface a t 4, 8, or 12 hr after inoculation with race 226 These treatments did not affect the growth ol infection hyphae and production of mature haustoria It is not feasible, therefore, that BcS inhibits production of certain fungal factors which elicit the resistance response in the host plant.Experiment 4: Effect of BcS on cross-protection was examined (Table 3) The abaxial sur- face of Shokan 1 leaves was first inoculated with incompatible race 226, and 20 hr later the adaxial surface of the same leaves was inoculated with compatible race 203 The development of race 203 was measured 48 hr later I n leaves without BcS treatment the length of infection hyphae and production of mature haustoria of normally compatible second inoculum 203 became similar to those of the incompatible race 226 apparently due to cross-protection effect. How-
Table 3 Development of normally compatible race 203 of P coronata avenue in Shokan l oat leaves which were treated with BcS and then pre-inoculated with incompatible race 226
-- -- -
Development of second inoculum
- --
First Second Length of infection Mature haustorium Treatment inoculum inoculum hyphae ( P ) (% of total haustoria)
Water 203 BcS 203 Watcr 226 BcS 226 Water 226 203 BcS 226 203
BcS (5 x M) was supplied for 4 hr prior to the first inoculation Sccond inoculation was made 20 hr after the first inoculation Developmcnt of the second inoculum was recorded 48 hr after the second inoculation.
100 Tech. Bull.. Fac. Agr. Kagawa Univ ever, in the leaves treated with BcS bcfore the first inoculation, both hyphal length and number of mature haustoria were identical to those of race 203 which was inoculated alone Thus BcS appears to block cross-protective response
Experiment 5: Antifungal activity in the leaf exudate and the crude preparation fiorn Shokan 1 leaves inoculated with race 226 was assayed. As shown in Table 4, the antifungal activity to germ tube elongation of race 226 was detected in both the leaf exudate and the crude preparation from inoculated leaves, but it was not detected in those from BcS-treated inoculated leaves.
Table 4 Germ tube growth of race 226 of P coronata avenue on aqueous solutions of the leaf exudate and thc crude preparation from Shokan 1 oat leaves which were inoculated with incompatible race 226 and then treated with BcS
-
Condition of leaf sample Germ tube length ( p ) on Inoculum Treatment Leaf exudate Crude preparation
Water 514 49 7
226 Watcr 26 1 217
BcS 538 494
226 BcS 522 51 1
BcS (5 x M) was supplied at 12 hr after inoculation for 4 hr, and the leaf exudate and the crude preparation were prepared at 48 hr after inoculation
The results in Experiments 4 and 5 may implicate possible contribution of BcS in repressing the resistance through its action to the host plant
Discussion
A number of antimetabolites and plant growth regulators have been used in the studies of host- pathogen interaction in rust diseases(4~8,11~12~14,15), I n these studies, however, emphasis has
been mostly laid on inducing resistance in normally susceptible host plants So far as we are aware, only ethylene(2) and maleic hydrazide(l0) are known to be effective in reducing resistance of wheat to Puccznza gramznzs trztzci and P recondzta, respectively This is probably due, at least in part, to difficulty in finding chemicals satisfactory to reverse the plants from resistant to sus- ceptible.
Previous attempts to produce uredospores of incompatible race 226 on oat leaves using nine plant growth regulators including ethylene and maleic hydrazide, six analogues of purine and pyrimidine bases, and six amino acid analogues were unsuccessful (unpublished). However, in the following experiments, we used protein synthesis inhibitors acromycin, BcS, chloramphenicol, cycloheximide, erythromycin, kasugamycin, and puromycin and found that BcS and puromycin a t concentrations of 5
x
10-6- 10-5 M and 2 5 x 10-4- 5 x 10-4 M, respectively, allowed to produce considerable number of urcdosori of incompatible race 226 It appears, therefore, that reversion of the rust infection type to susceptible by chemicals should not be achievcd by non- specific reduction of physiological activity of host plants but by the specific blockage of certain mechanism of resistance response Furthermore, the occurrence itself of such effective chemicals for the reversion of response may be a strong indication that resistance expression of the host plant requires certain de novo protein synthesis.Vol. 2 7, No. 59 (1976) 101 The effect of BcS on normally incompatible race 226 in Shokan 1 was remarkable for both vegetative and reproductive development of the fungus, since the time courses of hyphal growth, production of mature haustoria, and differentiation of uredia in BcS-treated leaves were identical to those of compatible race 203 (Figs 1 and 2-A, -B). BcS treatment also accelerated the hyphal growth of race 226 in resistant cultivar Bond (Table 1) and uredial differentiation in H K K which normally exhibited another type of resistance (Fig. 2-D) BcS, however, had no effect a t 5 x 10-6 M on the development of compatible race 203 in any oat cultivars (Figs. 1 and 2-C, Table 1) These results strongly suggest that BcS does not affect the susceptibility but represses the physiological process of resistance expression by the host. This presumption was supported by the facts in Table 3 that BcS completely inhibited the cross-protection response which is interpreted to involve defense reactions leading to the production of antifungal substance in the rust infections(ls6.7) including the crown rust of oat(5) Indeed, the increase in anti- fungal activity in Shokan 1 leaves following infection with race 226 was repressed by BcS treat- ment (Table 4). I n addition, we could not obtain any fact supporting the possibility that BcS acted through its direct effect upon the fungus (Fig 3, Experiments 2 and 3).
Involvement, at least in part, of induced defense reactions of the oat plant to non-pathogenic rust fungi was also suggested in this study (Table 2). This is in accord with the contention of HEATH(^) and LEATH and ROWELL@) arising from the works with Uromyces phaseoli var uignae
and Puccinia graminzr, respectively, that limitation of non-pathogenic rust fungi is not caused by
nutrient shortage but by induced physiological reactions of non-host plants.
I n a subsequent paper, possible contribution of protein synthesis in the cultivar-specific resist- ance will be discussed based on the results using BcS.
Summary
The development of incompatible race 226 of Puccznia coronata auenae in primary leaves of oat
cultivars Shokan 1 and Hyuga-Kairyo-Kuro ceased 35-40 hr and 72-96 hr after inoculation, respectively, and no uredial differentiation occurred I n leaves of both cultivars treated with 5 x 10-6 M blasticidin S (BcS) prior to inoculation, however, the fungal development was identical to that of compatible race 203 producing a number of uredia. Similar effect of BcS on hyphal growth was observed in resistant cultivar Bond. Puccinia agropyri,
P.
recondita and Uromyces alopecuri, which were non-pathogenic to oat and produced no haustoria, producedhaustoria in BcS-treated leaves
Cross-protective response in Shokan 1 leaves induced by the first inoculation with incompatible race 226 was negated by the treatment with BcS before the first inoculation. BcS-treatment resulted in marked decrease in post-infectional antifungal activity in Shokan 1 leaves. There was no evidence that BcS stimulated directly the development of race 226
I t was indicated that BcS does not affect the susceptible infection but blocks the process of in- duced resistance in oat leaves to the crown rust fungus as well as to certain non-pathogenic rust fungi
Acknowledgements
The authors wish to thank Dr. K. TOMIYAMA (Nagoya University), Dr. Y. ASADA (Ehime University), Dr. T.. KUSAKA (Takeda Chemical Industry Co.. Ltd.), and Dr. H.. SUMI (Sankyo Co. Ltd..) for supply of antibiotics. This work was supported in part by Grant from the Ministry
Tech, Bull. Fac. Agr.. Kagawa Univ. of Education of Japan (Nos. 936004 and 976029).
Refesen
Effect of rust-inhibiting compounds on the metabolism of wheat leaves, Can J Bot.,
39, 1019-1027 (1961)
SHAW, M : Cell biological aspects of host- parasite relations of obligate fungal parasites, Can J Bot ,45, 1205-1220 (1967)
SIIIPION, W A , BROWN, J F : A whole-leaf clearing and staining technique to demonstrate host-pathogen relationship of wheat stem rust, Phytopathology, 52, 13 13 (1962).
TANI, T , IRIKURA, H , NAIIO, N : Inhibition of urcdosorus formation of Puccinza coronata by plant growth regulators and antimetabolites, Tech Bull Fac. Agr Kagawa Unzv, 23, 42-50 (1971)
TANI, T , NAIIO, N , ONOE, T : Structure requirement of maleic hydrazide upon inhibitory effect of uredosorus formation of Puccznia coronata, Ann Phytopathol Soc Japan, 38, 428-430 (1972) TANI, T , YAMAMOIO: H , ONOE, T., NAITO, N :
Initiation of resistance and host cell collapse in the hypcrsensitivc reaction of oat leaves against Puccznza coronata avenae, Phyrzol Plant Pathol, 7, 231-242
TANI, 'II , YOSHIKAWA, M , NAIIO; N : Tem- plate activity of ribonucleic acid extracted from oat leaves infected by Puccinza coronata, Ann Phytopathol Soc Japan, 39, 7-13 (19 73) YAMAGUCIII, H , YAMAMOIO, C., TANAKA, N.: Inhibition of protein synthesis by blasticidin S I Studies with cell-free systems from bacterial and mammalian cells, J Bzochem , 57, 667- 677 (1965)
YAMAMOIO, H , NAIIO, N
,
TANI, T., TAKE-UCHI, K.: Susceptibility of oats and gramine-
ous crops to Puccznza coronata f sp avenae race 226 found in Kagawa, Tech Bull. Fas. Agr. Kagawa Unzv ,24, 171-1 76 (1973) In Japanese with English summary
YAMAMOIO, H , TANI, T , NAIIO, N : Changes in protein contents of oat leaves during the resistant reaction against Puccznza coronata avenae, Plytopatho1 2 , 82, 138-145 (1975)
tion of resistance of wheat to stem rust, Tvanr Brzt Mycol. Soc
,
58, 333-336 (1972).DALY, J. M , SEEVERS, P M., LUDDER, P :
Studies on wheat stem rust resistance controlled at the Sr6 locus I11 Ethylene and disease reaction, Phytopathology, 60, 1648-1652 (1970) HEAIH, M. C.: Light and electron microscopic studies of the interactions of host and non-host plants with cowpea rust-Uromycar phareolz var. vzgnaa, Phyrzol Plant Pathol , 4, 403-414 (1974). I~EITEPUSS, R : Dcr Einfluss von Actinomycin auf Puccznza grarnznzr Lrztzcz auf Weizen und dcn Einbau von Ortsaure-C" und Uridin-Ha in Wirtspflanze und Parasit, Phylopathol 2 , 69, 107-114 (1970).
KADOIA, G
,
TANI, T , NAIIO, N : Isolation of antifungal substance from oat leavcs infected by the crown rust fungus, Ann Phytopathol Soc. Japan, 41, 258 (1975) Abstr. in Japanese LEATH, K T , ROWELL, J B : Nutritional and inhibitory factors in the resistance of Zaa nzayr to Puccznza gramzntr, Phytopa~lzology, 60,1097-1100 (1970)
LIITLEFIELD, L. J.: Flax rust resistance in- duced by prior inoculation with an avirulent race of Melantprora lznz, Phytopatlzology, 59, 1323-
1328 (1969)
NAITO, N
,
LEE, M , TANI, T.: Inhibition of germination and infection structure formation of Puccinia coronata uredospores by plant growth regulators and antimetabolites, Tech Bull Fac Agr. Kagawa Univ , 23, 51-56 (1971) NAIIO, N , TANI, T , ARAKI, T : Relation- ship between parasite developmcnt and infection type in oat, wheat, and barley inoculated with Puccznza coronata, Trans Mycol Soc Japan, 11,16-22 (1970)
SAMBORSKI, D J , PERSON, C , FORSY IH, F R :
Differential effects of maleic hydrazide on the growth of leaf and stem rusts of wheat, Can J B o t , 38, 1-7 (1960)
Vol. 27, No. 59 (1976)
Puccinia coronata avanae $%$a 1. -.x 226 4)%Bd&, I 7 r c P
a@%%
1 4 k h 7JB @3&%0% 1 %l.Ekb'Z,?h'i-"h%@35-40 G P d k h V72-96 @Pd%tEIS;d L., BE?,%??%@, L % h L h L, k %, @ I % f E 5 ~ 1 0 - ~ M 8 ~ Y f x 1..tt.1 9 7 S ( B c S ) , T @ E L T k ( 2 , E#%Et&%$~'k1.,-x203tR@EffI.=&ET, % % a BE?f@fi%%&Lf:,, R@hmr;iJ[&f&%&a"a@d?~ l $ l . Z k l / l , - C &%b bh?:. Puccinia agropyri, P.. recondzta kJ. V Uromyce,~ alopecuri ItBts, 7 r q P %$E&CC.@B%b& L h bfJ', BcS @B%,T d&%%??@&
L?:.
$%$nakl.-x 226 8 % 1 &@@l.E h 'T%g S $L &H%i 1-30 cross-protection E E t & % 1 & @ @ h Q % %tE BcS @@,$& t 3% L %dl-> f:,. -3. f: , @Z 1 4.35 td: t.,- 7: 226 ~ @ l ~ j i , B E f J i % & + b 7 8 , BcS %BT? 0 @&
~ t ~ m c m i u 5 WE.
-.z,
BCS ~ J $ E % V - . X 226 t z t m L T % B Q E B S - ~ ~ taqgcaa
r,n&fihT ?c..Hk.D%#.R;6.b, I. ?/$P%ii! V f i t Z k b T , BcS C & % $ ~ % 1 / ~ - ~ ~ a ) @ % t E l t % % ~ @ ~ , ;l;%$ti'B~-%%hV -.3a>W%Li3j%i! ZP'BO@AtEt$