西 南 交 通 大 学 学 报
第 54 卷 第 6 期
2019 年 12 月
JOURNAL OF SOUTHWEST JIAOTONG UNIVERSITY
Vol. 54 No. 6
Dec. 2019
ISSN: 0258-2724 DOI:10.35741/issn.0258-2724.54.6.15
Research Article Materials Science
E
FFECTS OF
L
IQUID
S
MOKE ON THE
C
HEMICAL
C
OMPOSITION AND
T
HERMAL
P
ROPERTIES OF
S
AGO
F
IBER
液体烟雾对萨戈纤维化学成分和热学性质的影响
Mukhlis Muslimin a,*, Kusno Kamil c, Sofyan Arif Setya Budi b, I. N. G. Wardana ba Department of Mechanical Engineering, Khairun University
Ternate, Indonesia, mukhlis@unkhair.ac.id
b
Department of Mechanical Engineering, Brawijaya University Malang, Indonesia, kusno.kamil@umi.ac.id
c Department of Mechanical Engineering, Universitas Muslim Indonesia
Makassar, Indonesia, sasbudi@ub.ac.id, wardana@ub.ac.id
Abstract
The chemical composition and thermal properties of sago fiber, as processed via liquid smoke treatment, were analyzed in the current study, which is divided into two steps: fiber treatment and chemical property analysis. In the first stage, groups of sago fiber were immersed in the liquid smoke solution for one, two, three, four, and five hours prior to one-hour of drying, while untreated sago fiber was kept as the control specimen. After immersion, each group of sago fiber specimen was identified through SEM-EDS, which indicated that the effect of liquid smoke treatment resulted from escalated thermal stability as well as activation energy (from 213.4 °C and 67.02 kJ/mol to 220.6 °C and 73.43 kJ/mol, respectively). Additionally, a statistical approach depicted improved chemical properties of the treated sago fiber and confirmed the applicability of liquid smoke immersion as an appropriate treatment for sago fiber, prior to their application as reinforced composites.
Keywords: Sago Fiber, Liquid Smoke, Chemical Property, Thermal Property
摘要 在本研究中,对通过液体烟气处理的西米纤维的化学成分和热性能进行了分析,分为两个步 骤:纤维处理和化学性能分析。 在第一步中,将一组西米纤维浸入烟液中一小时,两小时,三小 时,四小时和五小时,然后干燥一小时,而未处理的西米纤维则作为对照样品。 浸泡后,通过扫 描电镜鉴定每组西米纤维样品,表明液烟处理的效果是由热稳定性和活化能(从 213.4 C 和 67.02 千焦/摩尔升高到 220.6 C 和 73.43 千焦/摩尔)。另外,一种统计方法描述了处理过的西米纤维的 化学性能得到改善,并证实了液体烟浸作为西米纤维的合适处理方法的适用性,然后再将其用作 增强复合材料。 关键词: 西米纤维,液体烟雾,化学性质,热性质
I. I
NTRODUCTIONThere are several direct applications of conventional composites in the form of green composites, such as in sports equipment, building materials, aircraft parts, naval applications, and automobile parts. Natural fibers possess high specific tensile strength, entail low production cost, and have low density along with easy recyclability, all of which makes them attractive alternatives compared to other fiber composites [1], [2], [3], [4], [5], [6], [7], [8], [9], [10], [11], [12], [21].
The characteristics of these natural fibers, and hence their utilization, are known to vary as composite reinforcement materials for light load and do not require high strength as compared to composite reinforcing materials for high load applications with superior physical and mechanical properties [13], [14], [15], [16], [17], [19], [20]. Commonly, fiber treatment methods are inexpensive and environment friendly. Certain previous studies performed treatment methods such as immersion with solutions (NaOH, KMnO4, H2O2, and seawater), heating
with turmeric solution, and fumigation. However, such types of treatment lead to the formation of specific chemical compositions on each fiber material. Subsequently, the changes that occur should affect the fibers’ physical and mechanical properties.
Previous research asserts that soaking treatment of coco fiber with NaOH reduces lignin levels, while immersing such a fiber with KMnO4 and H2O2 should improve fiber grooves
[5]. Treatment with palm fiber by immersion in seawater provides more chemical compatibility to composite resin. Here, the lignin in the fibers is flushed out, making the fiber surface coarser with better bonds to the matrix [2], [3]. An alternative treatment of turmeric solution on Akaa midrib fiber also increases cellulose composition and lignin content of the fiber, so that the surface morphology becomes coarse and increases fiber strength [7]. Fiber fumigation of a king pineapple leaf is very influential on fiber surface morphology due to changes in chemical composition. It becomes coarser and more grooved, which improves compatibility between epoxy and the fibers [9]. The percentage of crystallinity and thermal stability were also found to be maximal in sisal-g-poly (MA) followed by benzoylated sisal fibers [1]. Some observations indicate that the texture of the treated fiber results from liquid smoke that has transformed the surface into a grooved and rugged surface. So the
bond between fiber and matrix becomes more compact [18].
As one of the studies that are environmentally concerned, this study applied liquid smoke as the solution agent with unique ingredients. Its hydrophilic characteristic contained OH groups including polar compositions with electronegativity, which compounded to fibers to improve fiber composition and strength. This research used sago fiber and immersion in liquid smoke obtained by an enrichment process method. The study expected to reveal changes in the chemical composition and thermal properties of the fibers, and in the end should increase the mechanical properties of the fibers.
II. M
ATERIAL ANDM
ETHODS A. MaterialThe raw materials used in this research were sago fiber (SF) from Luwu regency, South Sulawesi province, Indonesia and liquid smoke.
(a) sago palm (b) sago fibers Figure 1. Illustration of sago fibers Table 1.
Chemical components of liquid smoke
No Component Percentage
1 L-alanine, ethyl ester, 2-propanamine,1-methoxy 2-pentanamine
33.37
2 2, 3-butanedione, acetic acid ethyl ester
02.60
3 Acetic acid 56.65
4 Acetic acid, methyl ester 07.39
B. Methods
After being separated from midrib leaf, SF was immersed in a liquid smoke solution for specific durations, as shown in Table 2.
Table 2. Study notation
No Treatment Notation Remarks
1 No treatment TP No
immersion, no drying
43 0C for 1 hour 3 2 hours immersion P2J 4 3 hours immersion P3J 5 4 hours immersion P4J 6 5 hours immersion P5J
Groups of specimen were treated and coded as listed in Table 1. After immersion, the fiber was dried in an electric oven at 43 0C for one hour and then took out for slow cooling at room temperature.
C. SEM-EDS of SF
The morphology of the fiber surface of each group was then observed by the electron
microscope Vega3 Tescan Scanning Electron Microscope (SEM) at a 5 kV voltage.
D. Differential Scan Calorymetry (DSC) of SF
DSC is one of the Thermal Analyzer tools to determine heat capacity and components of a material. DSC is an analytical technique that measures calories in samples and serves as a comparator as a function of temperature.
III. R
ESULTS ANDD
ISCUSSION A. SEM-EDS ObservationFor the SEM-EDS test, the elements contained in the fiber with no treatment (TP) and with treatments (P1J-P5J) are shown in Figure 2.
Figure 2. SEM-EDS test results of SF (TP, P1J, P2J, P3J, P4J, and P5J)
The elements contained in each test are notated in Table 3.
Table 3.
The elements of SF resulted by SEM-EDS test
Element Oxide Sample SF (%)
TP P1J P2J P3J P4J P5J 2 4 6 8 10 12 14 keV 0.0 0.5 1.0 1.5 2.0 2.5 3.0 cps/eV Na Mg Al Si S S O K K Ca Ca TP 2 4 6 8 10 12 14 keV 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 cps/eV Na Mg Al Si P S S Ca Ca O P1J 2 4 6 8 10 12 14 keV 0 2 4 6 8 10 cps/eV Na Mg Al Si P S S O P2J 2 4 6 8 10 12 14 keV 0 200 400 600 800 1000 x 0.001 cps/eV Na Mg Al Si P S S O P3J 2 4 6 8 10 12 14 keV 0.0 0.2 0.4 0.6 0.8 1.0 1.2 cps/eV Na Mg Al Si P S S O P4J 2 4 6 8 10 12 14 keV 0.0 0.2 0.4 0.6 0.8 1.0 1.2 1.4 cps/eV Na Mg Al Si P S S O P5J
Sodium Na2O 16.42 15.37 15.17 17.56 11.85 12.71 Magnesium MgO 16.47 14.75 14.05 14.29 8.83 12.54 Aluminium Al2O3 16.71 15.62 24.24 34.58 13.18 9.97 Silicon SiO2 22.40 26.42 25.66 14.49 54.21 34.68 Phosphorus P2O3 0 15.45 11.32 9.34 4.87 19.08 Sulfur SO3 15.02 12.1 9.56 9.74 7.07 11.04 Calcium CaO 9.22 0.29 0 0 0 0 Potassium K2O 3.77 0 0 0 0 0
From the data in Table 3, the relationship between the content of the elements in SF with the time of treatment can be seen. Test results found that the composition of phosphorus plays an important role in the formation of fiber compounds. Before treatment, the fiber did not contain phosphorus, but after the treatment of P1J-P5J, phosphorus increased to 15.45, 11.32, 9.34, 4.87, and 19.08%, respectively. Meanwhile, the calcium element degraded from 9.22% before treatment to just 0.29% after 1 hour of treatment. After treatment P2J-P5J, no calcium remained in the fiber. This means changes in the chemical
composition of the fiber contributed to changes to the fiber’s physical properties.
B. Differential Scanning Calorimetry (DSC) Observation
DSC functions as a Thermal Analyzer and is used to determine the heat and enthalpy capacity of a specimen. It determines the delta H and exothermal area, which affects the heat release process in the glass transition area. From the test results, it was found that the thermal capacity of the fibers with more exothermal areas had more carbon in the fiber. The test results are shown in Figure 3.
a. No treatment (TP)
b. Treatment 1 hour (P1J) Eksothermal 1
Eksothermal 1
c. Treatment 2 hours (P2J) Eksothermal 2 Eksothermal 1
d. Treatment 3 hours (P3J) Eksothermal 2 Eksothermal 1
e. Treatment 4 hours (P4J) Eksothermal 2 Eksothermal 1
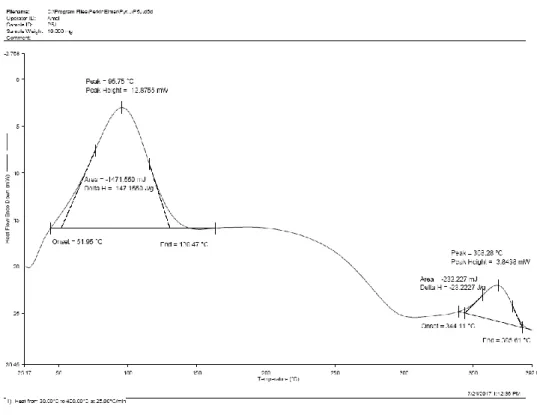
f. Treatment 5 hours (P5J)
Figure 3. Result of DSC – exothermal peak (TP, P1J, P2J, P3J, P4J, dan P5J)
Exothermic events in the TP samples spanned an area of 54.80 to 139.97 degrees, which were onset by exothermic peaks at 102.08 0C and and subsequently burned out. Exothermal areas in the TP sample only occurred once because H2O
levels remained dominant in the fiber and because of the weak bonds of carbon elements.
DSC measurements evaluated the influence of liquid smoke on the latent heat capacity of the SF. The SF without treatment had a latent heat capacity of 102.08-degree Celsius. While treated fiber P1J indicated the latent heat capacity occurred at two areas, the first area decreased to 99.45 0C and the second increased at 364.92 0C. In experiments P1J-P5J, there were two
exothermal areas for each due to the reduction of H and O elements and the increase of the element C’s degree of polymerization (DP) whenever treated with liquid smoke.
The results of Perkin Elmer DSC tests showed that the untreated (TP) and treatment (P1J-P5J) fibers had differences in exothermal area. In fibers without treatment, the exothermal area only occurred once, namely: area = -2236.857 mJ, delta H = -223.6857 J/g, peak = 102.08 0C, and peak height = - 17.7476 mW, at the exothermal temperature starting at 54.80 0C and ending at 139.97 0C. Whereas in fibers with the exothermal treatment area it occurs in 2 areas, as shown in the following Table 4.
Table 4.
Result DSC (TP, P1J, P2J, P3J, P4J, dan P5J) Eksothermal 1
No. Fiber sample
Eksothermal 1
Onset End Area Delta H Peak Peak height 0 C 0C mJ J/g 0C mW 1 TP 54.8 139.97 -2236.9 -223.69 102.08 -17.7476 2 P1J 59.01 134.27 -1351.6 -135.16 99.45 -11.7602 3 P2J 59.87 141.82 -1557.3 -155.73 105.28 -12.6679 4 P3J 60.33 142.07 -1393.2 -139.32 106.1 -11.2861 5 P4J 48.17 125.83 -1438.7 -143.87 90.76 -12.7648 6 P5J 51.95 130.47 -1471.6 -147.16 95.75 -12.8755 No. Fiber sample Eksothermal 2
Onset End Area Delta H Peak Peak height 0 C 0C mJ J/g 0C mW 1 TP 0 0 0 0 0 0 2 P1J 339.31 385.05 -261.36 -26.136 364.92 -3.9432 3 P2J 342.51 388.17 -273.83 -27.383 369.56 -4.0791 4 P3J 343.1 385.53 -127.52 -12.752 369.12 -2.0519 5 P4J 339.58 387.2 -461.08 -46.108 367.92 -6.6103 6 P5J 344.11 385.61 -232.23 -23.223 368.28 -3.8438
In the chemical process of immersion, liquid smoke forms liquid-smoke fiber compounds. The heating process will degrade the composition of
H and O while forming carbon bonds within fiber compounds.
a. The process of H and O elements degradation of in lignin compounds
b. The process of H and O elements degradation in hemicellulose compounds
Before After
After Before
c. The process of H and O elements degradation in cellulose compounds Figure 4. The process of H and O elements degradation in SF compounds
IV. C
ONCLUSION1. There are changes in proportion of chemical composition of each treatments, indicating chemical reactions of liquid smoke compounds to the sago fiber.
2. Changes in exothermic and endothermal areas in fibers for each treatment show that the reactions between liquid smoke and sago fibers formed strong chemical bonds that affect the element of fiber compounds, which ended in changes of the physical and mechanical properties of the fiber.
3. The thermal stability and activation energy of the SF improved from 213.4 °C to 220.6 °C and 67.02 kJ/mol to 73.43 kJ/mol, respectively, due to liquid smoke treatment.
A
CKNOWLEDGMENTThis study is partly supported by Kemenristekdikti through the scheme of BUDI-DN Scholarship LPDP 2016 to 2019 in term of providing financial supports for this study.
R
EFERENCES[1] KALIA, S., KAUSHIK, V.K., and
SHARMA,
R.K.
(2011)
Effect
of
Benzoylation and Graft Copolymerization on
Morphology,
Thermal
Stability,
and
Crystallinity of Sisal Fibers. Journal of
Natural Fibers, 8 (1), pp. 27-38.
[2] MARDIN, H., WARDANA, I.N.G,
SUPRAPTO, W., and KAMIL, K. (2016)
Effect of Sugar Palm Fiber Surface on
Interfacial Bonding with Natural Sago Matrix.
Advance
in
Material
Science
and
Engineering, 7, pp. 1-5.
[3] AHSAN, Q., CARRON, T.S.S., and
MUSTAFA, Z. (2019) On the use of nano
fibrillated
kenaf
cellulose
fiber
as
reinforcement
in
polylactic
acid
biocomposites.
Journal
of
Mechanical
Engineering and Sciences, 13 (2), pp.
4970-4988.
[4] MARDIN, H., WARDANA, I.N.G.,
KUSNO, K., PRATIKTO, and WAHYONO,
S. (2016) Sea Water Effects on Surface
Morphology and Interfacial Bonding of
Sugar Palm Fiber to Sago Matrix. Key
Engineering Materials, 724, pp. 39-42.
[5] ARSYAD, M., WARDANA, I.N.G,
PRATIKTO, and IRAWAN, Y.S. (2015) The
morphology of coconut fiber surface under
chemical treatment. Revista Materia, 20 (1),
pp. 169-177.
[6] ARSYAD, M., WARDANA, I.N.G,
PRATIKTO, and IRAWAN, Y.S. (2015)
Bonding ability of coconut fiber with
polyester matrix as a result of chemical
treatment. International Journal of Applied
Engineering Research, 10 (4), pp. 9561-9570.
[7]
RENRENG,
I.,
SOENOKO,
R.,
PRATIKTO, and IRAWAN, Y.S. (2017)
Effect of Turmeric (Curcuma Longae)
Treatment on Morfologi and Chemical
properties of Akaa (Corypha) Single Fiber.
Journal of Engineering Science and
Technology, 12 (8), pp. 2229-2237.
[8] PALUNGAN, M.B., SOENOKO, R.,
IRAWAN, Y.S., and PURNOWIDODO, A.
(2017) Structure Crystallization of Cellulose
King Pineapple Leafs Fiber (Agave Cantala
Roxb) Due to Smoke Fumigation. ARPN
Journal of Engineering and Applied Sciences,
12 (20), pp. 5721-5725.
[9] PALUNGAN, M.B., SOENOKO, R.,
IRAWAN, Y.S., and PURNOWIDODO, A.
(2016) The Effect of Fumigation Toward the
Engagement Ability of King Pineapale Leaf
Fibre (Agave Cantala Roxb) with Epoxy
Matrix. ARPN Journal of Engineering and
Applied Sciences, 11 (13), pp. 8532-8537.
[10] CHAFIDZ, A., RIZAL, M., RM, F.,
KAAVESSINA, M., HARTANTO, D., and
ALZAHRANI, S.M. (2018) Processing and
After Before
Hydrogen Oxygen Carbon