つくばリポジトリ Po 12 12 e0190333
全文
図
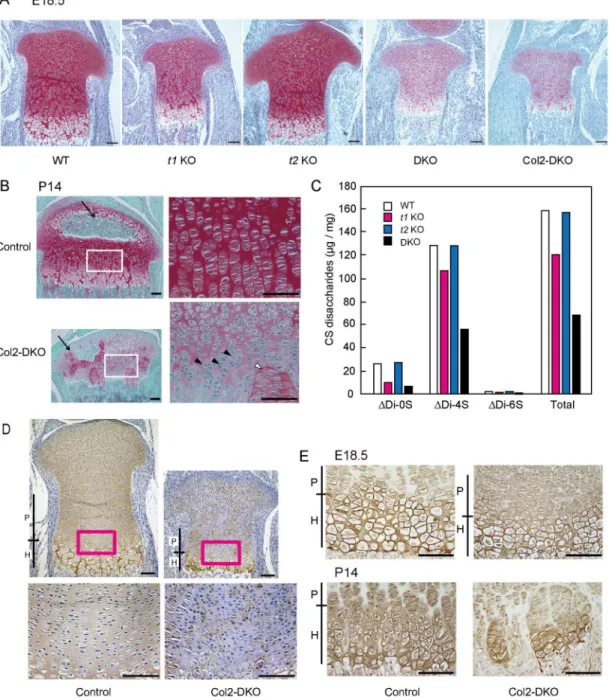
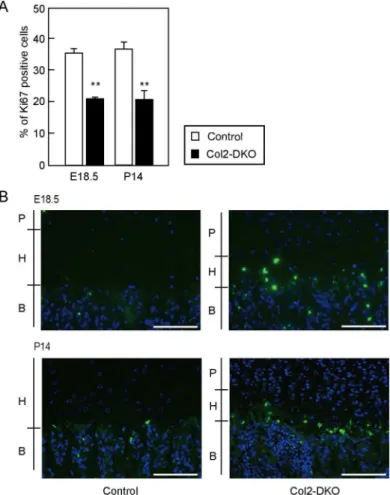
関連したドキュメント
The tumor suppressor Reck is critical for vascular patterning and stabilization in mice( Dissertation_全文 ). Glicia, Maria
The mGoI framework provides token machine semantics of effectful computations, namely computations with algebraic effects, in which effectful λ-terms are translated to transducers..
An example of a database state in the lextensive category of finite sets, for the EA sketch of our school data specification is provided by any database which models the
A NOTE ON SUMS OF POWERS WHICH HAVE A FIXED NUMBER OF PRIME FACTORS.. RAFAEL JAKIMCZUK D EPARTMENT OF
A lemma of considerable generality is proved from which one can obtain inequali- ties of Popoviciu’s type involving norms in a Banach space and Gram determinants.. Key words
If in the infinite dimensional case we have a family of holomorphic mappings which satisfies in some sense an approximate semigroup property (see Definition 1), and converges to
These results are motivated by the bounds for real subspaces recently found by Bachoc, Bannai, Coulangeon and Nebe, and the bounds generalize those of Delsarte, Goethals and Seidel
de la CAL, Using stochastic processes for studying Bernstein-type operators, Proceedings of the Second International Conference in Functional Analysis and Approximation The-