Tech Bull Fac Agr Kagawa Univ, Vol 46, No 2, 101-107, 1994
CRYOPRESERVATION OF IN VITRO-GROWN DIANTHUS
BY ENCAPSULATION-DEHYDRATION..
Seiichi FUKAI, Mariko TOGASHI a n d Masanori GOI
Nodal segments of zn vttro-grown Dtanthus hybrtdus cv Sakuranadesiko were cryopreserved by the two-step freezing and the encapsulation-dehydration methods Drought tolerance was induced in alginate encapsulated explants by preculture on medium enr~ched with 0 2-1 OM sucrose The encapsulated explants, precultured and dried to about 20% water by weight, showed 100% survival and shoot regeneration following immersion in liquid nitrogen Explants cryopreserved by conventional two-step freezing showed lower shoot regeneration
Key words : cryopreservation, Dtanthus, encapsulation, dehydration
Introduction
T h e genus Dzanthus consists of about 300 species T h e s e a r e a n important genetic resource for introduction of n e w characteristics t o t h e modern carnation cryopreservation of shoot tips of Dzanthus species grown in a greenhouse has already been developed by using t h e two-step freezing method('*2)
Recently, alginate-encapsulation, which w a s developed a s t h e synthetic seed, has been successfully adapted for cryopreservation of plant germplasm(3) This method encapsulates explants in alginate beads T h e s e beads are precultured on medium containing high level of sucrose, then partially dried prior to immersion in LN2 Successful cryopreservation by this encapsulation-dehydration method has been reported in several species(33435s6)
In this paper, w e compare encapsulation-dehydration with t h e conventional two-step freezing method for cryopreservation of in vitro grown Dzanthus s t e m segments
Materials and Methods
Plant materzals
.
In vztro shoot cultures of dianthus (Dzanthus hybzrdus c v Sakuranadesiko) were maintained on medium containing 3 g / l Hyponexe (complete soluble fertilizer, N-P-K=6-65-19, Hyponex Co L t d Ohio), 20g/l sucrose and 8g/l agar a t 25°C with a light level of 19 ,umol/sec/m2 provided by cool white fluorescent tubes for 16hr/day T h e s e plants were subcultured every 6 weeks by nodal cuttings For cryopreservation experiments, explants (ca 2mm s t e m segments containing one node without leaf) were excised from nodes positioned first and second from top on t h e 4 to 6 weeks old shoots with fully expanded leavesAbbreviations : BA-6-benzylaminopurine, DMSO-dimetyl sulfoxide, LN2 -liquid nitrogen, MS-Murashige &
102 Tech. Bull. Fac. Agr. Kagawa Univ., Vol. 46, No.2, 1994
Two-step freezing method
:
Explants were put into Iml plastic straws (bull semen straws, Fujihira Industry Co. Ltd.) with cryoprotectant solution containing 10% DMSO and 3% glucose, and incubated for 1 hour at 0°C. T h e straws were then cooled a t a rate of 0.5"C/min. to -40'C in a programmable freezing apparatus (Program freezer ET-1, Fujihira Industry Co. Ltd.). When the temperature of the cooling bath had reached -3.5"C, the straws were briefly touched the middle part of t h e straw with tweezers that had been cooled in LN2 to induce ice crystal formation. T h e temperature of the cooling bath w a s then held a t - 5 C for l0min. During gradual cooling, some straws were thawed and others were then immersed into LN2 a t various temperatures. T h e frozen straws were thawed rapidly in a water bath (25-30°C). Thawed explants were rinsed three times with CPW salts s o ~ u t i o n ' ~ ' and cultured on the shoot tip culture medium containing half strength of M S salts and vitamins"', O.lmg/l BA, 0.5mg/l NAA, 20g/l sucrose and 8g/l agar ( S T medium).Fig. 1 Encapsulated explants (beads)
Encapsulation : Excised explants were suspended in S T medium without CaC12 and supplemented with 3% sodium alginate (100-150cP). Drops containing one explant were then pipeted into lOOmM CaC12 solution. After 30min the beads (Fig. 1) were removed from t h e solution.
Induction of drought tolerance : T h e beads were precultured on S T medium containing 0-1.OM sucrose for 2 days. T h e beads were then air-dried for several hours in t h e lamina flow cabinet. A t various points during this drying process, the weight of some equivalent dummy beads w a s measured, after which these beads were dried in a oven (105°C) for 16h to calculate water content of the beads a t each point in the drying process.
Cryopreservation of encapsulated explants
:
T h e beads, precultured and partially dried, were put in 2ml cryo-tubes and immersed directly in L N 2 After 15min. immersion, the tubes were thawed rapidly in a water bath (25-30°C). Thawed beads were then cultured on S T medium.S FUKAI et a1 : Cryopreservation of Dtanthus by encapsulation-dehydration
within 8 weeks of culture
Results 1 Cryopreservation by two-step freezing method
Explants, cooled gradually to various temperatures and thawed without immersion into LN2,
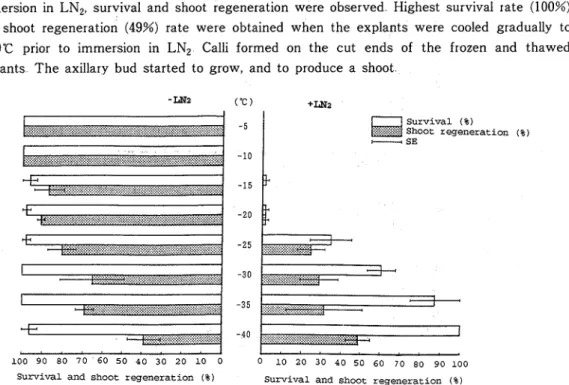
showed high survival rates, but shoot regeneration decreased a s the explants were cooled a t lower temperatures (Fig 2) Explants cooled gradually to above -20°C, followed by immersion in LN2, showed nearly zero% survival In explants cooled gradually to below -25°C prior to immersion in LN2, survival and shoot regeneration were observed Highest survival rate (100%) and shoot regeneration (49%) rate were obtained when the explants were cooled gradually to -40°C prior to immersion in LN2 Calli formed on the cut ends of t h e frozen and thawed explants T h e axillary bud started to grow, and to produce a shoot
Survival ( % )
Shoot regeneration ( % )
-
SEI-'
100 90 80 7 0 60 50 40 30 20 10 0
Survival and shoot regeneration (8)
~ - -
0 10 20 30 40 SO 60 70 80 90 100
Survival and shoot regeneration ( % )
Fig 2 Survival and shoot regeneration of explants cooled gradually to various temperature
Each value represents the average of 3 replicates of 15 explants
2 Cryopreservation by encapsulation-dehydration
Viability of explants dried under air flow declined sharply when water content was reduced below 50% of weight regardless of encapsulation (Fig 3-A,
B)
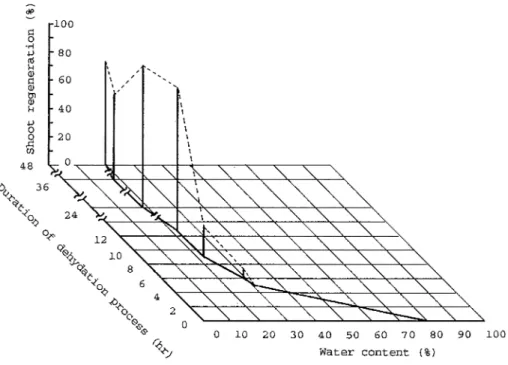
Explants encapsulated and precultured for 2 days showed high drought tolerance, except for explants precultured on medium free of sucrose (Fig 3-C, D) During drying, the size of beads reduced remarkably a t lower concentrations of sucrose in the preculture medium Dried beads recovered their original size in one day after culture on S T mediumT h e beads, that were precultured on medium containing 0 4--1 OM sucrose then dried to about 20% water by weight prior to immersion in LN2, showed a 100% rate of survival and shoot regeneration (Fig 4) T h e beads that were precultured on medium containing 0 2M
Tech Bull Fac Agr Kagawa Univ , Vol 46, No 2, 1994 100: 0 0 0 90 : o
A
80:
70 : 0 60 : 50:
4 0 : 30 : 20 7 0lo
: 0 0 " " " " " " " " I 100 90 80 7 0 60 50 40 30 20 1 0 0 Water content ( % ) I00 r 0 0 ~ 0 o 0 oB
90:
80-
70:
60:
50:
o 4 0:
o 30 7 20 7 10 : 0 0 1 0 0 90 80 7 0 60 50 40 30 20 10 0 water content ( % ) water content ( % ) 100-
03 0 0 00ocaxxDD
90 : 80:
70-
60-
50:
40 7 30 : 20 : 10-
0 " " " ~ ' . ' . ' . ' - s ~ ~ ~ I 1 0 0 90 80 7 0 60 50 40 30 20 1 0 0 Water content ( % ) Fig 3 Drought tolerance of explantsA : intact explants
B : encapsulated explants without preculture
C
.
encapsulated explants precultured on the medium free of sucroseD ' encapsulated explants precultured on the medium containing 0 2-1 OM
sucrose
S. FUICAI et al. : Cryopreservation of Dianthus by encapsulation-dehydration.
0 10 20 3 0 4 0 5 0 60 7 0 8 0 9 0 100
Water content ( % )
Fig. 4 Shoot regeneration of encapsulated explants after direct immersion in LN2. Encapsulated explants were precultured on the medium containing 0.6M sucrose for 2 days, partially dried to various percentages of water by weight prior to immersion in LN2.
Fifteen explants were used in each treatment.
Fig. 5 Shoot regeneration of encapsulated explants frozen in LN2.
sucrose showed lower shoot regeneration (max. 60%). No survival of explants precultured on medium free of sucrose was observed.
Water content of t h e beads w a s critical for survival of the explants after immersion in LN2. No survival w a s observed when beads had more than 32% water by weight. Sufficiently low water content w a s obtained after air drying for 11 to 13 hr in the lamina flow cabinet. Drying for longer, however, resulted in a drop in the rates of survival and shoot regeneration. After they thawed, encapsulated explants turned white in one day. Only axillary buds recovered their original green color, and regenerated shoots (Fig. 5).
Tech Bull Fac Agr Kagawa Univ , Vol 46, No 2, 1994
Discussion
Most drought tolerant seeds can survive following direct immersion in LN2 if their water content is between 5 to 10% of weight(g) Residual water in seeds vitrifies instead of forming ice crystals, this results in serious mechanical damage to their cells Cryopreservation of partially dried tissue('0) and somatic embryos(") has already been successfully achieved Control of water content in the diminutive explant is, however, difficult when they are dried under ambient conditions Encapsulation-dehydration has been explored to solve this problem(3)
Our results demonstrate that encapsulation does not induce drought tolerance in explants (Fig 3-B), although encapsulated explants, precultured on medium enriched with sucrose, show high drought tolerance (Fig 3-D) Accumulation of sucrose has been reported when drought tolerance was induced in pollen('2) Once drought tolerance is induced, the encapsulated explants, dried to about 20% water by weight survive direct immersion in LN2 While the explants are drying, sucrose in the beads is concentrated enough to vitrify when cooled rapidly('3) These results suggest that encapsulation may act a s a buffer to prevent too rapid dehydration of explants by trapping sucrose
Explants frozen by the conventional two-step freezing method, however, show significantly lower shoot regeneration Shoot tips of Dzanthus species excised from mother plants grown in a greenhouse show nearly 100% survival and shoot regeneration rates when frozen in the two-step freezing r n e t h ~ d ( ~ " ~ ) T h e lower shoot regeneration documented in this experiment may be because stem explants are comparatively larger than shoot tips (2mm vs 0 5mm) Our results show that lateral buds suffer serious damage below -25°C However, it is necessary to reach temperature below -25°C to adequately dehydrate the tissue, thus ensuring survival following immersion in LN2
In conclusion, stem explants of zn vztro grown dianthus have been successfully cryopreserved
when encapsulated with alginate, precultured on medium containing 0 4-0 1M sucrose and partially dried to 20% water by weight This technique would be applicable to any species of genus Dzanthus which can be maintained in vitro
Acknowledgement
We thank Stacey Iseman for her help in preparing the manuscript
References (1) UEMURA, M and SAKAI, A : Survival of carnati-
on (Dtanthus caryophyllus L ) shoot apices frozen to the temperature of liquid nitrogen
Plant Cell Phystol , 21, 85-94 (1980)
(2) FUKAI, S , GOI, M and TANAKA, M : Cryoprese- rvation of shoot tips of Caryophyllaceae ornamentals Euphttca, 56, 149-153 (1991) (3) FABRE, J and DEREUDDRE, j : Encapsulation-
dehydration : A new approach to cryopreserva- tion of Solanum shoot tips Cryo-Lett , 11,
413-426 (1990)
(4) NIINO, T and SAKAI, A : Cryopreservation of alginate-coated in vitro-grown shoot tips of apple, pear and mulberry Plant Scz, 87,
199-206 (1992)
(5) PLESSIS, P , LEDDET, C and DEREUDDRE, J :
Resistance to dehydration and to freezing in liquid nitrogen of alginate-coated shoot-tips of grape vine (Vttts vtntfera L cv Chardonnay) A R Aacd Sct Parts, t 313 Ser 111, 373-380
S F U K A ~ et a l : Cryopreservation of Dtanthus by encapsulation-dehydration 107 (1991)
(6) PAULET F , ENGELMANN, F , and GLAZMANN J-C : Cryopreservation of apices of tn vttro plantlets of sugarcane (Saccharurn s p hybrids) using encapsulation/ dehydration Plant Cell Reports, 12, 525 -529 (1993)
(7) POWER, J B and CHAPMAN, J V : Plant cell culture Isolation, culture and genetic manipu- lation of plant protoplasts I n R A Dixon (ed ), Plant cell culture p 37-66 IRL Press, Oxford (1985)
(8) MURASHIGE, T SKOOG, F : A revised medium for rapid growth and bioassays with tobacco tissue cultures Phystol P l a n t , 15, 473-497 (1962)
(9) STANWOOD, P C : Cryopreservation of seed germplasm for genetic conservation I n K K Kartha (ed), Cryopreservation of plant cells and organs pp 199-226 CRC Press, Florida (1985)
(10) URAGAMI, A , SAKAI, A and NAGAI, M :
Cryopreservation of dried axillary buds from plantlets of Asparagus offtctnalts L grown in
vitro Plant Cell Report, 9, 328-331 (1990)
UD SHIMONISHI, K , ISHIKAWA, M , SUZUKI, S and OOSAWA, K : Cryopreservation of melon somatic embryos by desiccation method J a p a n J Breed, 41, 347-351 (1991)
(12) HOEKSTRA, F A and VAN ROEKEL, T : Desic- cation tolerance of Papaver dubtum L pollen during its development in the anther Plant Phystol , 88, 626-632 (1988)
(13) DEREUDDRE, J , HASSEN, N , BLANDIN, S and ,
KAMINSKI, M : Resistance of alginate-coated somatic embryos of carrot (Daucus carota L ) to desiccation and freezing in liquid nitrogen 2 Thermal analysis Cr yo-Letters, 12 135-148 (1991)
(14) DREEUDDRE, J , FABRE, J and BASSAGLIA, C :
Resistance to freezing in liquid nitrogen of carnation (Dtanthus caryophyllus L var Eolo) apical and axillary shoot tips excised from different aged in vitro plantlets Plant Cell Reports, 7, 170-173 (1988)