Tech Bull Fac Agr Kagawa Univ Vol 30, No 63, 19-44 1978
T H E RELATION BETWEEN T H E SILAGE QUALITY AND
T H E REDUCTION
OF NITRATE DURING ENSILAGE
Mitsuaki OHSHIMA, Kiyotoshi OOUCHI and Noboru KUKINO
Two experiments were carried out to study the effects of glucose supplementation and density of herbage in silo upon the nitrate reduction and the nitrogen oxides gas production during ensilage. For the purpose, 300, 400, 500 and 600g of' chopped ladino clover were packed into experimental silos with or without glucose so a s to occupy 800 cm3.
The addition of glucose improved the silage quality and depressed the nitrate reduction. The untreated silages attained pH values of arround 4.6 and contained 2 to 3 % of the original nitrate in both the two experiments. T h e glucose treated silages were similar in their p H values (arround 3.9) but different in the reduction rates of nitrate between the two experi- ments. They were 95% in Experimentt 1 and 53 to 77% in Experiment 2 showing negative relation to the density of herbage in silo which had no obvious effect on the silage quality. More nitrogen oxides gas was recovered from the treated silages in spite of less reduction of nitrate during ensilage than from the untreated ones in both experiments and the recovery was increased according t o the increase of the density of herbage in silo in both the treated and untreated silages of Experiment 2.
9 4 ~ - . ~ % \ & @ D @ @ D ~ ~ I Z ~ A I E ~ - ~ - 4 L - V E ~ Q ) % @ ~ , 2 H(Z,H:Q&3 Lf:.
%$lcmBc%!%Lf:4V/ P D , - / ~ , - % ? ~ D $ $ , &tj4>624%%Q ~ % O I ~ J ~ ~ - X B % ~ J ~ J L ? : D S , 800ml%D
a n m s % - . @ r r 3 0 0 ( s @ 2 0361, 400, 500
;f;w
~ O O ~ T T Q - . C L ~ ~ A / - ~ ~ . ~ ~ 9 4U G H
~ c ~ i ~ ~ ~ ~L'f:.. %khkIZ.'#fd 7T%&,3'6 33 X G2 10M-KOH @KIBlR
L?:..
@ @ ~ + t b B l l h ? : . &iffj~o*4 v - 9 0 pH $s&Ll$jj~/ . i - h d z Q h 4 . 6 ; f ; k b 2 f J L \ L 3 % ~ & - 3 f : . . %jJa+f41/,-~
1/--g&ga
@Efd%C2fd: <, (r\Phb pH4 &5t>G2.i- hUTT&7f:CCbfilkl;h G 4 , @@E$%DBR%C2%L <%fd3f:,, ? j L b % . %@I TG2;hyfi,5%T&7f:D iC$j L, %@2 T i t 23 fLr/\ L 47% fiflE!@Shf:.. @~LP%~~x%[email protected]~BW@DB~;E~:G~ZIC, $331+f4 L.-Vfisz'4ta %fin94 - uBZ.#;Lg <, %f:%@2Tdd, S ~ & b B D @ f J a t r Lf:k%\, ~ # @ . @ C D @ ~ @ @ $ ~ K & T ~ ~ ~ ~ : ~ + I C D @iLS%D3J&fis%jJu Lf:,,Introduction
I t is known that nitrate in herbage is reduced to nitrite('), ammoniac2) and nitrogen oxides gad2) during ensilage and the rate of the reduction is dependent on the silage quality(3). A herbage containing nitrate in a high concentration is toxic to animals''). Therefore, the
20 Mitsuaki OHSHIMA, Kiyotoshi OOUCHI and Noboru KUKINO Tech Bull Fac. Agr Kagawa Univ.
reduction of nitrate during ensilage is one of the useful characteristics of silage fermentation. The object of the present study was to examine in depth the relationship between the reduction rate of nitrate and the silage quality, which had not been shown in detail a s yet. For the purpose, two experiments were carried out and a lot of experimental silos were prepared filling different volumes of ladino clover (Tr(foZinm repens L., ladino type) with or without glucose. These treatments were selected to prepare a number of silages different in quality.
Experimental Preparation of sillages
Ladino clover harvested on 27th May (1st cut, Experiment 1) and 15 t h July (2 nd cut, Experiment 2) 1976 was chopped to about 1 cm length using a straw chopper. Reagent bottles of 800 ml capacity were filled with 300 (only in Experiment 21, 400, 500 and 600 g of the chopped clover with or without the addition of glucose a t the rate of 2g/100g fresh herbage. Duplicate silos were prepared for each treatment. Each silo was sealed with a rubber stopper fitted with a fermentation trap containing 50 ml 10M-KOH to absorb gases and maintain anaerobic condition a s described pr e v i ~ u s l y ( ~ ) . The ensiling period was 90 days.
Analysis
Analysis of pH, dry matter (DM), total nitrogen (TN), volatile basic nitrogens (VBN), lactic acid and volatile fatty acids (VFA) were carried out using fresh samples. Nitrate and nitrite were determined using samples dried a t 70°C with a forced-air oven. For comparison, some fresh silage samples were also used for determining their nitrate contents and i t was found that there was no difference between the fresh- and dry-samples. The nitrogen oxides gas absorbed in the alkaline solution was determined immediately after detaching the trap from the silo. The methods other than those detailed below have been described in earlier publicationsc5). DM content was determined by the toluene distillation methodc6). T N content was determined by the improved Kjeldahl method recommended by AOAC for nitrate-containing samples('). Nitrate and nitrite were determined according to the method of AOAC(8). For comparison the method of MORRIS and GONZALEZ-M~S(~) was also used and found to be not suitable for the determination of nitrate in silage. Nitrogen oxides trapped into the alkaline solution were determined by the AOAC method for nitrate analysis using neutralized diluted solution.
Results
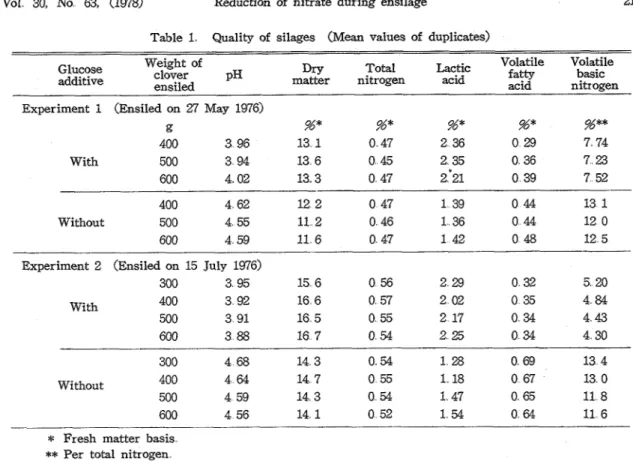
The quality of the silages is shown in Table 1. There was little or no relationship between the density of herbage in silo and the silage quality but considerable difference in fermentation characteristics was found between the silages ensiled with glucose and those ensiled without glucose in both experiments. By being added with glucose a t the rate of 2
%
of' the fresh material a t the time of ensiling, the silages of low pH value, high lactic acid content, low VFA and VBN contents were obtained and they were classified as excellent in quality. While the silages ensiled without glucose were of pH values arround 4.6 and contained less lacticVol 30, NO 63, (1978) Reduction of nitrate during ensilage 21 Table 1. Quality of silages (Mean values of duplicates)
Weight of
Dry Total Lactic Volatile fatty Vo basic
er pH matter nitrogen acid
led acid nitrogen
Experiment 1 (Ensiled on 27 May 1976)
g %* %* %* %* %** 400 3 96 13 1 0 47 2 36 0 29 7 74 With 500 3 94 13 6 0 45 2 35 0 36 7 23 600 4 02 13 3 0 47 2 "21 0 39 7 52 400 4 62 12 2 0 47 1 39 0 44 13 1 Without 500 4 55 11 2 0 46 1 36 0 44 12 0 600 4 59 11 6 0 47 1 42 0 48 12 5 Experiment 2 (Ensiled on 15 July 1976)
300 3 95 15 6 0 56 2 29 0 32 5 20 With 400 3 92 16 6 0 57 2 02 0 35 4 84 500 3 91 16 5 0 55 2 17 0 34 4 43 600 3 88 16 7 0 54 2 25 0 34 4 30 300 4 68 14 3 0 54 1 2 8 0 69 13 4 Without 400 4 64 14 7 0 55 1 18 0 67 13 0 500 4 59 14 3 0 54 1 47 0 65 11 8 600 4 56 14 1 0 52 1 54 0 64 11 6
*
Fresh matter basis**
Per total nitrogenacid and more VFA and VBN compared with the glucose-treated silages and classified a s modarate in quality.
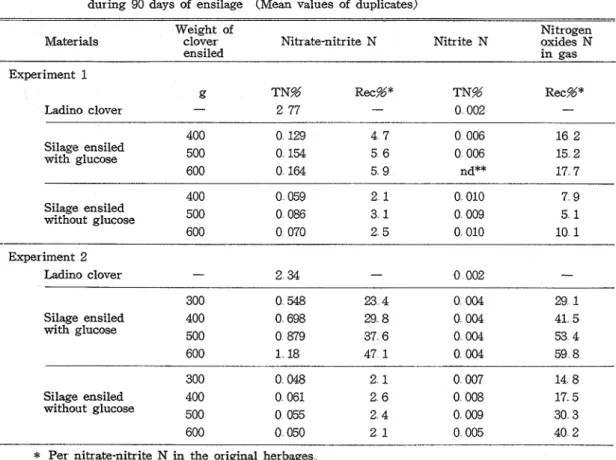
Nitrate and nitrite in both the herbages and silages and nitrogen oxides trapped into the alkaline solution were determined according to the method of AOAC. The results a r e shown in Table 2.
In Experiment 1, 5 and 16
5%
of' nitrate-nitrite N of the herbage were recovered in the glucose-treated silages and in their gaseous fractions, respectively. The untreated silages con- tained only about a half amount of nitrate-nitrite N but produced less nitrogen oxides gas compared with the treated ones. The density of herbage in silo had little or no effects on nitrate and nitrite contents of the silages and nitrogen oxides gas production during ensilage. While, in Experiment 2, the recovery of nitrate and the nitrogen oxides gas production of the treated silages were greater than those obtained in Experiment 1 and they were 35 and 46%,
respectively, in average of eight silos involving four treatments different in the density. The untreated silages in Experiment 2 showed similar nitrate content but higher nitrogen oxides gas production compared with those in Experiment 1. As is evident from Figure 1, in the treated silages of Experiment 2, the increase in the density of herbage in silo was accompanied with the increases of the recovery of nitrate in the silages and the nitrogen oxides gas production during ensilage. While in the untreated silages the density had no relation to the nitrate content of the silages but had possible relation to the nitrogen oxides gas production.22 Mitsuaki OHSHIMA, Kiyotoshi OOUCHI and Noboru KUKINO Tech Bull Fac Agr Kagawa Univ
Table 2 Nitrate and nitrite in herbages and silages and nitrogen oxides recovered from silos during 90 days of ensilage (Mean values of duplicates)
-Weight of Nitrogen
Materials clover Nitr ate-nitr ite N Nitrite N pxides N
ensiled In gas Experiment 1 g TN% Re&* TN% Rec%* Ladino clover
-
2 77-
0 002 - 400 0 129 4 7 0 006 16 2 Silage ensiled with glucose 500 0 154 5 6 0 006 15 2 600 0 164 5 9 nd** 17 7 400 0 059 2 1 0 010 7 9 Silage ensiled without glucose 500 0 086 3 1 0 009 5 1 600 0 070 2 5 0 010 10 1 Experiment 2 Ladino clover-
2 34 - 0 002-
300 0 548 23 4 0 004 29 1 Silage ensiled 400 0 698 29 8 0 004 41 5 with glucose 500 0 879 37 6 0 004 53 4 600 1 18 47 1 0 004 59 8 300 0 048 2 1 0 007 14 8 Silage ensiled 400 0 061 2 6 0 008 17 5 without glucose 500 0 055 2 4 0 009 30 3 600 0 050 2 1 0 005 40 2*
Per nitrate-nitrite N in the original herbages**
Not determinedWeight of herbage ensiled
Figure 1 The effect of density of herbage in silo on nitrate and nitrite contents (-) and nitrogen oxides gas production (-
-
-) of ladino clover silage ensiled with ( 0 ) or without ( 0 ) glucose in Experiment 2Vol. 30, No. 63, (1978) Reduction of nitrate during ensilage
Discussion
T h e data in Table 1 show that between Experiments 1 and 2, t h e quality of silages was quite similar within treatments in spite of being ensiled ladino clover different in stage. T h e silages treated with glucose showed excellent chemical quality while those ensiled without glucose showed moderate quality. But the reduction r a t e of nitrate in the treated silages was far larger in Experiment 1 than in Experiment 2 a s shown in Table 2. This result may have reflected a slight difference of silage quality. From t h e VBN content, the treated silages of Experiment 2 were considered to be a little superior in quality to those of Experiment 1. T h e nitrate contents of the untreated silages agreed well between t h e two experiments and smaller than those of the treated silages. I t is known t h a t nitrate in herbage decreases during ensilage by being reduced by some bacteria(lO), the r a t e being greater in poor silages than in lactate silages.(') In a previous report(5), the quality of silages was improved with the increase of herbage ensiled. While in t h e present experiments, obvious effect of t h e packing r a t e of herbage on the silage quality could not be observed. But the r a t e of nitrate- nitrite N to T N in the treated silages of Experiment 2 increased according to t h e increase in the density of herbage in silo. These results may have been derived from the different kinds of fermentation which should be enhanced and depress the activity of aerobic bacteria by packing the herbage tightly. ATAKA e t al.") reported t h a t a large part of nitrate in herbage was reduced a t t h e first stage of ensilage process by aerobic bacteria and the further reduction could not be observed. While BOUSSET e t ad. (lo) reported that besides Enterobacter sp.,
Lactobaczlus p l a n t a r u m which is dominant in t h e early stages of ensilage and Clostrzdzum tyrobutyrzcum could reduce nitrate. I t is also known t h a t Clostridzum sporogenes("), a dominant clostridia in butyrate silage, and Clostrzdzum welchii(12) reduce nitrate. HENDERSON and MCDONALD~'~) observed that recoveries of nitrate in ensiled ryegrass over 3 days were 75% in spite of t h e attainment of a low pH values(4.3). In t h e same experiment after 139 days, the silages contained 13 to 44
%
of the original nitrate. These results show that t h e reduction of nitrate during ensilage could be carried out by anaerobic bacteria besides aerobic organisms. In t h e present experiments, there observed negative relationship between t h e disappearance of nitrate from silage and the appearance of nitrogen oxides gas. From the fact, i t is considered t h a t in silages of good quality t h e smaller amount of nitrate was reduced to nitrite and most of the nitrite was lost from the silo a s gas, while in silages of poorer quality, the more amount of nitrate was reduced to nitrite and a large part of t h e nitrite was reduced to ammonia. T h e sum of nitrate-nitrite N and nitrogen oxides N of a silage packed the glucose-treated herbage most tightly exceeded t h e nitrate-nitrite N content of t h e original herbage suggesting the oxidation of nitrogen during ensilage. I t was observed t h a t some air leaked into the silos through the fermentation traps. But this may be a phenomenon unique to the small-scale laboratory silos. T h e same trends have been shown in another study(14).I t is concluded t h a t the addition of available carbohydrate a t t h e time of ensiling w a s effective in improving the legume silage quality but i t depressed the reductions of nitrate and nitrite during ensilage. By increasing the density of herbage in silo from 300 to 600 g/800
24 Mitsuaki OHSHIMA, Kiyotoshi OOUCHI and Noboru KUKINO Tech Bull Fac Agr. Kagawa Univ
cm3, the silage quality was little effected but the reduction of nitrate was depressed. References
(1 ) ATAKA, K., NARASAKI, N., KIKUCHI, M , MATSUI, Y : Proc 31st Ann Meet Jap Soc Grass1 Sci., 95 (1976)
( 2 ) No, E , YOSHIDA, J , NAKAMURA, R : ibid., 94 (1976)
( 3 ) JACOBSON, W C., WISEMAN, H G : J. Dairy S c i , 46, 617 (1963)
( 4 ) MIYAZAKI, A : Jap J Zootech Sci., 48,
53 (1977).
( 5 ) OHSHIMA, M., AKAZAWA, M., TANAKA, H.: Tech Bull Fac. Agric. Kagawa Univ., 25, 191 (1974).
( 6 ) DEWAR , W A , MCDONALD, P : J. Sci Fd Amz'c., 12, 790 (1961).
( 7 ) Association of Official Analytical Chemists: Official Methods of Analysis, 11th ed., The Association, Washington, 126 (1970)
( 8 ) Association of Official Analytical Chemists: i b i d , 17 (1970)
( 9 ) MORRIS, M P., GONZALEZ-MAS, A : J Agr Food Chem., 6 , 456 (1958)
(10) BOUSSET-FATIANOFF, N , GOUET, Ph., BOUSSET, J , CONTREPOIS, M : Ann Biol anim Biochem Biophys, 1 1 , 715 (1971)- (11) LAMANNA, C , MALLETTE, M F.: Basic
Bacteriology, 3 rd ed , Wiliams and Wilkins Co , Baltimore, 844 (1965)
(12) HASAN, S M , HALL, J B : J gen Micro- biol. 87, 120 (1975)
(13) HENDERSON, A R , MCDONALD, P : J Sci Fd Agric., 22, 157 (1971)
(14) OHSHIMA, M , MCDONALD, P , ACAMOVIC, T : i b i d , unpublished